Claudin-1 induced sealing of blood-brain barrier tight junctions ameliorates chronic experimental autoimmune encephalomyelitis
- PMID: 21983942
- PMCID: PMC3207130
- DOI: 10.1007/s00401-011-0883-2
Claudin-1 induced sealing of blood-brain barrier tight junctions ameliorates chronic experimental autoimmune encephalomyelitis
Abstract
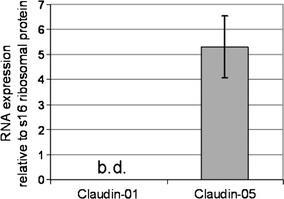
In experimental autoimmune encephalomyelitis (EAE), an animal model for multiple sclerosis (MS), loss of the blood-brain barrier (BBB) tight junction (TJ) protein claudin-3 correlates with immune cell infiltration into the CNS and BBB leakiness. Here we show that sealing BBB TJs by ectopic tetracycline-regulated expression of the TJ protein claudin-1 in Tie-2 tTA//TRE-claudin-1 double transgenic C57BL/6 mice had no influence on immune cell trafficking across the BBB during EAE and furthermore did not influence the onset and severity of the first clinical disease episode. However, expression of claudin-1 did significantly reduce BBB leakiness for both blood borne tracers and endogenous plasma proteins specifically around vessels expressing claudin-1. In addition, mice expressing claudin-1 exhibited a reduced disease burden during the chronic phase of EAE as compared to control littermates. Our study identifies BBB TJs as the critical structure regulating BBB permeability but not immune cell trafficking into CNS during EAE, and indicates BBB dysfunction is a potential key event contributing to disease burden in the chronic phase of EAE. Our observations suggest that stabilizing BBB barrier function by therapeutic targeting of TJs may be beneficial in treating MS, especially when anti-inflammatory treatments have failed.
Figures



References
-
- Coisne C, Dehouck L, Faveeuw C, Delplace Y, Miller F, Landry C, Morissette C, Fenart L, Cecchelli R, Tremblay P, Dehouck B. Mouse syngenic in vitro blood-brain barrier model: a new tool to examine inflammatory events in cerebral endothelium. Lab Invest. 2005;85:734–746. doi: 10.1038/labinvest.3700281. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

