Tumor biomarker glycoproteins in the seminal plasma of healthy human males are endogenous ligands for DC-SIGN
- PMID: 21986992
- PMCID: PMC3270097
- DOI: 10.1074/mcp.M111.008730
Tumor biomarker glycoproteins in the seminal plasma of healthy human males are endogenous ligands for DC-SIGN
Abstract
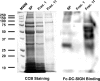
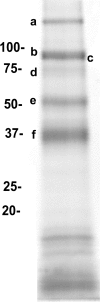
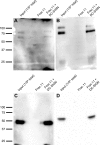
DC-SIGN is an immune C-type lectin that is expressed on both immature and mature dendritic cells associated with peripheral and lymphoid tissues in humans. It is a pattern recognition receptor that binds to several pathogens including HIV-1, Ebola virus, Mycobacterium tuberculosis, Candida albicans, Helicobacter pylori, and Schistosoma mansoni. Evidence is now mounting that DC-SIGN also recognizes endogenous glycoproteins, and that such interactions play a major role in maintaining immune homeostasis in humans and mice. Autoantigens (neoantigens) are produced for the first time in the human testes and other organs of the male urogenital tract under androgenic stimulus during puberty. Such antigens trigger autoimmune orchitis if the immune response is not tightly regulated within this system. Endogenous ligands for DC-SIGN could play a role in modulating such responses. Human seminal plasma glycoproteins express a high level of terminal Lewis(x) and Lewis(y) carbohydrate antigens. These epitopes react specifically with the lectin domains of DC-SIGN. However, because the expression of these sequences is necessary but not sufficient for interaction with DC-SIGN, this study was undertaken to determine if any seminal plasma glycoproteins are also endogenous ligands for DC-SIGN. Glycoproteins bearing terminal Lewis(x) and Lewis(y) sequences were initially isolated by lectin affinity chromatography. Protein sequencing established that three tumor biomarker glycoproteins (clusterin, galectin-3 binding glycoprotein, prostatic acid phosphatase) and protein C inhibitor were purified by using this affinity method. The binding of DC-SIGN to these seminal plasma glycoproteins was demonstrated in both Western blot and immunoprecipitation studies. These findings have confirmed that human seminal plasma contains endogenous glycoprotein ligands for DC-SIGN that could play a role in maintaining immune homeostasis both in the male urogenital tract and the vagina after coitus.
Figures

References
-
- Garcia-Vallejo J. J., van Kooyk Y. (2009) Endogenous ligands for C-type lectin receptors: the true regulators of immune homeostasis. Immunol. Rev. 230, 22–37 - PubMed
-
- Geijtenbeek T. B., Torensma R., van Vliet S. J., van Duijnhoven G. C., Adema G. J., van Kooyk Y., Figdor C. G. (2000) Identification of DC-SIGN, a novel dendritic cell-specific ICAM-3 receptor that supports primary immune responses. Cell 100, 575–585 - PubMed
-
- Appelmelk B. J., Van Die I., Van Vliet S. J., Vandenbroucke-Grauls C. M., Geijtenbeek T. B., Van Kooyk Y. (2003) Cutting edge: carbohydrate profiling identifies new pathogens that tnteract with dendritic cell-specific ICAM-3-grabbing nonintegrin on dendritic cells. J. Immunol. 170, 1635–1639 - PubMed
-
- Mitchell D. A., Fadden A. J., Drickamer K. (2001) A novel mechanism of carbohydrate recognition by the C-type lectins DC- SIGN and DC-SIGNR: Subunit organization and binding to multivalent ligands. J. Biol. Chem. 276, 28939–28945 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

