Divergent synthesis and chemical reactivity of bicyclic lactone fragments of complex rearranged spongian diterpenes
- PMID: 21988207
- PMCID: PMC3202687
- DOI: 10.1021/ja207727h
Divergent synthesis and chemical reactivity of bicyclic lactone fragments of complex rearranged spongian diterpenes
Abstract
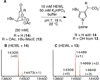
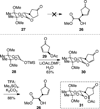
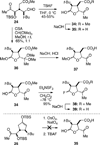
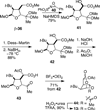
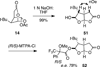
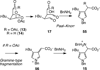
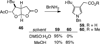
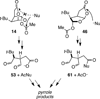
The synthesis and direct comparison of the chemical reactivity of the two highly oxidized bicyclic lactone fragments found in rearranged spongian diterpenes (8-substituted 6-acetoxy-2,7-dioxabicyclo[3.2.1]octan-3-one and 6-substituted 7-acetoxy-2,8-dioxabicyclo[3.3.0]octan-3-one) are reported. Details of the first synthesis of the 6-acetoxy-2,7-dioxabicyclo[3.2.1]octan-3-one ring system, including an examination of several possibilities for the key bridging cyclization reaction, are described. In addition, the first synthesis of 7-acetoxy-2,8-dioxabicyclo[3.3.0]octanones containing quaternary carbon substituents at C6 is disclosed. Aspects of the chemical reactivity and Golgi-modifying properties of these bicyclic lactone analogs of rearranged spongian diterpenes are also reported. Under both acidic and basic conditions, 8-substituted 2,7-dioxabicyclo[3.2.1]octanones are converted to 6-substituted-2,8-dioxabicyclo[3.3.0]octanones. Moreover, these dioxabicyclic lactones react with primary amines and lysine side chains of lysozyme to form substituted pyrroles, a conjugation that could be responsible for the unique biological properties of these compounds. These studies demonstrate that acetoxylation adjacent to the lactone carbonyl group, in either the bridged or fused series, is required to produce fragmented Golgi membranes in the pericentriolar region that is characteristic of macfarlandin E.
Figures








Similar articles
-
Golgi-modifying properties of macfarlandin E and the synthesis and evaluation of its 2,7-dioxabicyclo[3.2.1]octan-3-one core.Proc Natl Acad Sci U S A. 2010 Apr 6;107(14):6158-63. doi: 10.1073/pnas.1001421107. Epub 2010 Mar 23. Proc Natl Acad Sci U S A. 2010. PMID: 20332207 Free PMC article.
-
Versatile Construction of 6-Substituted cis-2,8-Dioxabicyclo[3.3.0]octan-3-ones: Short Enantioselective Total Syntheses of Cheloviolenes A and B and Dendrillolide C.J Am Chem Soc. 2017 May 31;139(21):7192-7195. doi: 10.1021/jacs.7b04265. Epub 2017 May 22. J Am Chem Soc. 2017. PMID: 28514145
-
General Access to Concave-Substituted cis-Dioxabicyclo[3.3.0]octanones: Enantioselective Total Syntheses of Macfarlandin C and Dendrillolide A.J Org Chem. 2020 Dec 4;85(23):15532-15551. doi: 10.1021/acs.joc.0c02273. Epub 2020 Nov 16. J Org Chem. 2020. PMID: 33197184
-
Spongian diterpenoids from marine sponges.Nat Prod Rep. 2006 Apr;23(2):321-34. doi: 10.1039/b503531g. Epub 2006 Feb 20. Nat Prod Rep. 2006. PMID: 16572231 Review.
-
[Construction of All-cis-substituted Cyclopropane and Total Synthesis of Avenaol].Yakugaku Zasshi. 2019;139(10):1259-1265. doi: 10.1248/yakushi.19-00130. Yakugaku Zasshi. 2019. PMID: 31582609 Review. Japanese.
Cited by
-
Covalent modification of biological targets with natural products through Paal-Knorr pyrrole formation.Nat Prod Rep. 2017 Aug 30;34(9):1051-1060. doi: 10.1039/c7np00024c. Nat Prod Rep. 2017. PMID: 28808718 Free PMC article. Review.
-
Investigation of the electrophilic reactivity of the biologically active marine sesquiterpenoid onchidal and model compounds.Beilstein J Org Chem. 2018 Aug 24;14:2229-2235. doi: 10.3762/bjoc.14.197. eCollection 2018. Beilstein J Org Chem. 2018. PMID: 30202476 Free PMC article.
-
Synthetic and Biological Studies of Sesquiterpene Polygodial: Activity of 9-Epipolygodial against Drug-Resistant Cancer Cells.ChemMedChem. 2015 Dec;10(12):2014-26. doi: 10.1002/cmdc.201500360. Epub 2015 Oct 5. ChemMedChem. 2015. PMID: 26434977 Free PMC article.
-
Synthesis of N-alkoxycarbonyl Pyrroles from O-Substituted Carbamates: A Synthetically Enabling Pyrrole Protection Strategy.J Org Chem. 2023 Oct 6;88(19):13584-13589. doi: 10.1021/acs.joc.3c01257. Epub 2023 Sep 20. J Org Chem. 2023. PMID: 37729493 Free PMC article.
-
Bioactive micronutrients in coffee: recent analytical approaches for characterization and quantification.ISRN Nutr. 2014 Jan 22;2014:384230. doi: 10.1155/2014/384230. eCollection 2014. ISRN Nutr. 2014. PMID: 24967266 Free PMC article. Review.
References
-
-
For reviews see Keyzers RA, Northcote PT, Davies-Coleman MT. Nat. Prod. Rep. 2006;23:321. González M. Cur. Bioact. Comp. 2007;3:1. Faulkner DJ. Nat. Prod. Rep. 2001;18:1.
-
-
- Molinski TF, Faulkner DJ, He CH, Van Duyne GD, Clardy J. J. Org. Chem. 1986;51:4564.
-
- Bobzin SC, Faulkner DJ. J. Nat. Prod. 1991;54:225. - PubMed
-
- Dumdei EJ, Dilip de Silva E, Andersen RJ, Choudhary MI, Clardy J. J. Am. Chem. Soc. 1989;111:2712.
- Morris SA, Dilip de Silva E, Andersen RJ. Can. J. Chem. 1991;69:768.
- Rungprom W, Chavasiri W, Kokpol U, Kotze A, Garson MJ. Marine Drugs. 2004;2:101.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

