Varicella zoster virus ORF25 gene product: an essential hub protein linking encapsidation proteins and the nuclear egress complex
- PMID: 21988664
- PMCID: PMC3230707
- DOI: 10.1021/pr200628s
Varicella zoster virus ORF25 gene product: an essential hub protein linking encapsidation proteins and the nuclear egress complex
Abstract
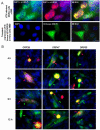
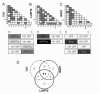
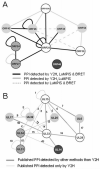
Varicella zoster virus (VZV) ORF25 is a 156 amino acid protein belonging to the approximately 40 core proteins that are conserved throughout the Herpesviridae. By analogy to its functional orthologue UL33 in Herpes simplex virus 1 (HSV-1), ORF25 is thought to be a component of the terminase complex. To investigate how cleavage and encapsidation of viral DNA links to the nuclear egress of mature capsids in VZV, we tested 10 VZV proteins that are predicted to be involved in either of the two processes for protein interactions against each other using three independent protein-protein interaction (PPI) detection systems: the yeast-two-hybrid (Y2H) system, a luminescence based MBP pull-down interaction screening assay (LuMPIS), and a bioluminescence resonance energy transfer (BRET) assay. A set of 20 interactions was consistently detected by at least 2 methods and resulted in a dense interaction network between proteins associated in encapsidation and nuclear egress. The results indicate that the terminase complex in VZV consists of ORF25, ORF30, and ORF45/42 and support a model in which both processes are closely linked to each other. Consistent with its role as a central hub for protein interactions, ORF25 is shown to be essential for VZV replication.
Figures

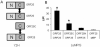
Similar articles
-
Characterization of the Varicella-zoster virus ORF25 gene product: pORF25 interacts with multiple DNA encapsidation proteins.Virus Res. 2009 Sep;144(1-2):58-64. doi: 10.1016/j.virusres.2009.03.019. Epub 2009 Apr 7. Virus Res. 2009. PMID: 19720242 Free PMC article.
-
Intermolecular Complementation between Two Varicella-Zoster Virus pORF30 Terminase Domains Essential for DNA Encapsidation.J Virol. 2015 Oct;89(19):10010-22. doi: 10.1128/JVI.01313-15. Epub 2015 Jul 22. J Virol. 2015. PMID: 26202238 Free PMC article.
-
The Varicella-zoster virus DNA encapsidation genes: Identification and characterization of the putative terminase subunits.Virus Res. 2007 Nov;129(2):200-11. doi: 10.1016/j.virusres.2007.07.015. Epub 2007 Sep 14. Virus Res. 2007. PMID: 17868947 Free PMC article.
-
Glycoproteins of varicella-zoster virus and their herpes simplex virus homologs.Rev Infect Dis. 1991 Nov-Dec;13 Suppl 11:S960-3. doi: 10.1093/clind/13.supplement_11.s960. Rev Infect Dis. 1991. PMID: 1664135 Review.
-
Intracellular synthesis of varicella-zoster virus.J Infect Dis. 1992 Aug;166 Suppl 1:S7-12. doi: 10.1093/infdis/166.supplement_1.s7. J Infect Dis. 1992. PMID: 1320653 Review.
Cited by
-
Changes in subcellular localization reveal interactions between human cytomegalovirus terminase subunits.Virol J. 2012 Dec 21;9:315. doi: 10.1186/1743-422X-9-315. Virol J. 2012. PMID: 23259714 Free PMC article.
-
Structure of Epstein-Barr virus tegument protein complex BBRF2-BSRF1 reveals its potential role in viral envelopment.Nat Commun. 2020 Oct 26;11(1):5405. doi: 10.1038/s41467-020-19259-x. Nat Commun. 2020. PMID: 33106493 Free PMC article.
-
Sensitivity of the C-Terminal Nuclease Domain of Kaposi's Sarcoma-Associated Herpesvirus ORF29 to Two Classes of Active-Site Ligands.Antimicrob Agents Chemother. 2018 Sep 24;62(10):e00233-18. doi: 10.1128/AAC.00233-18. Print 2018 Oct. Antimicrob Agents Chemother. 2018. PMID: 30061278 Free PMC article.
-
Mutual Interplay between the Human Cytomegalovirus Terminase Subunits pUL51, pUL56, and pUL89 Promotes Terminase Complex Formation.J Virol. 2017 May 26;91(12):e02384-16. doi: 10.1128/JVI.02384-16. Print 2017 Jun 15. J Virol. 2017. PMID: 28356534 Free PMC article.
-
The varicella-zoster virus portal protein is essential for cleavage and packaging of viral DNA.J Virol. 2014 Jul;88(14):7973-86. doi: 10.1128/JVI.00376-14. Epub 2014 May 7. J Virol. 2014. PMID: 24807720 Free PMC article.
References
-
- Mettenleiter TC, Klupp BG, Granzow H. Herpesvirus assembly: an update. Virus Res. 2009;143(2):222–34. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

