The application of carbon nanotubes in target drug delivery systems for cancer therapies
- PMID: 21995320
- PMCID: PMC3210734
- DOI: 10.1186/1556-276X-6-555
The application of carbon nanotubes in target drug delivery systems for cancer therapies
Abstract
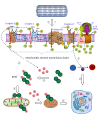
Among all cancer treatment options, chemotherapy continues to play a major role in killing free cancer cells and removing undetectable tumor micro-focuses. Although chemotherapies are successful in some cases, systemic toxicity may develop at the same time due to lack of selectivity of the drugs for cancer tissues and cells, which often leads to the failure of chemotherapies. Obviously, the therapeutic effects will be revolutionarily improved if human can deliver the anticancer drugs with high selectivity to cancer cells or cancer tissues. This selective delivery of the drugs has been called target treatment. To realize target treatment, the first step of the strategies is to build up effective target drug delivery systems. Generally speaking, such a system is often made up of the carriers and drugs, of which the carriers play the roles of target delivery. An ideal carrier for target drug delivery systems should have three pre-requisites for their functions: (1) they themselves have target effects; (2) they have sufficiently strong adsorptive effects for anticancer drugs to ensure they can transport the drugs to the effect-relevant sites; and (3) they can release the drugs from them in the effect-relevant sites, and only in this way can the treatment effects develop. The transporting capabilities of carbon nanotubes combined with appropriate surface modifications and their unique physicochemical properties show great promise to meet the three pre-requisites. Here, we review the progress in the study on the application of carbon nanotubes as target carriers in drug delivery systems for cancer therapies.
Figures




References
-
- Ferrari M. Cancer nanotechnology: opportunities and challenges. Nat Rev Cancer. 2005;3:161–171. - PubMed
-
- Cui D, Zhang H, Sheng J, Wang Z, Toru A, He R, Tetsuya O, Gao F, Cho H-S, Cho S, Huth C, Hu H, Pauletti GM, Shi D. Effects of CdSe/ZnS quantum dots covered multi-walled carbon nanotubes on murine embryonic stem cells. Nano Biomed Eng. 2010;2(4):236–244.
-
- Huang P, Zhang C, Xu C, Bao L, Li Z. Preparation and characterization of near-infrared region absorption enhancer carbon nanotubes hybridmaterials. Nano Biomed Eng. 2010;2(4):225–230.
-
- Bao C, Tian F, Estrada G. Improved visualisation of internalised carbon nanotubes by maximising cell spreading on nanostructured substrates. Nano Biomed Eng. 2010;2(4):201–207.
-
- Chen D, Wu X, Wang J, Han B, Zhu P, Peng Ch. Morphological observation of interaction between PAMAM dendrimer modified single walled carbon nanotubes and pancreatic cancer cells. Nano Biomed Eng. 2010;2(4):60–65.
LinkOut - more resources
Full Text Sources
Other Literature Sources

