Nuclear Ago2/HSP60 contributes to broad spectrum of hATSCs function via Oct4 regulation
- PMID: 21995449
- PMCID: PMC3261027
- DOI: 10.1089/ars.2011.4134
Nuclear Ago2/HSP60 contributes to broad spectrum of hATSCs function via Oct4 regulation
Retraction in
-
Retraction of S-K. Kang et al., articles in ARS.Antioxid Redox Signal. 2012 Sep 1;17(5):848. doi: 10.1089/ars.2012.1540. Antioxid Redox Signal. 2012. PMID: 22746834 Free PMC article.
Abstract
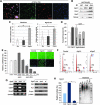
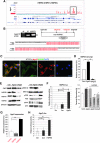
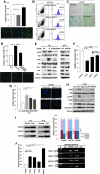
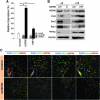
Aims: Argonaute2 (Ago2) plays a fundamental role in microRNA-mediated gene regulation through its intrinsic endonuclease activity. In this study we demonstrate the novel functions and molecular mechanisms by which nuclear Ago2 directly regulates HSP (heat shock protein) 60 expression and stem cell self-renewal. HSP60 is a crucial regulator of ROS (reactive oxygen species), senescence, and apoptotic cell death in several tissues and cell types.
Results: HSP60 is regulated via inactivation of p38/JNK and p53 and binds directly to the regulatory regions of the TERT, c-myc, GPx3, p53, and STAT3 genes. Using HSP60 CHIP-PCR experiments, we show that HSP60 binds directly to the Oct4 and Nanog genes and directly regulates Oct4 and other stemness genes involved in human adipose tissue-derived stem cell (hATSC) differentiation. HSP60 also positively regulates ROS-scavenging factors, including GPx3 and TXNL1, which directly modulate cytosolic ROS in hATSCs. Moreover, our study shows that Oct4 regulates HSP60 expression and controls hATSC survival and self-renewal after binding to the HSP60 gene. Furthermore, HSP60-mediated regulation of Oct4 contributes to neuronal and endodermal β-cell differentiation of hATSCs in vitro and in vivo and downregulates mesoderm-specific gene expression.
Innovation and conclusion: We show that increased levels of Ago2 or HSP60 effectively induce nuclear localization of HSP60, which directly controls Oct4, c-Myc, p53, TERT, and STAT3 for transdifferentiation programs. Collectively, we suggest a novel model in which nuclear Ago2 controls HSP60 in hATSCs.
Figures




Similar articles
-
MBD6 is a direct target of Oct4 and controls the stemness and differentiation of adipose tissue-derived stem cells.Cell Mol Life Sci. 2013 Feb;70(4):711-28. doi: 10.1007/s00018-012-1157-4. Epub 2012 Sep 30. Cell Mol Life Sci. 2013. PMID: 23052207 Free PMC article.
-
Crucial role of nuclear Ago2 for hUCB-MSCs differentiation and self-renewal via stemness control.Antioxid Redox Signal. 2012 Jan 15;16(2):95-111. doi: 10.1089/ars.2011.3975. Epub 2011 Sep 21. Antioxid Redox Signal. 2012. Retraction in: Antioxid Redox Signal. 2012 Sep 1;17(5):848. doi: 10.1089/ars.2012.1540. PMID: 21902595 Retracted.
-
Nuclear Argonaute 2 regulates adipose tissue-derived stem cell survival through direct control of miR10b and selenoprotein N1 expression.Aging Cell. 2011 Apr;10(2):277-91. doi: 10.1111/j.1474-9726.2011.00670.x. Aging Cell. 2011. PMID: 21241449
-
Oct4 expression revisited: potential pitfalls for data misinterpretation in stem cell research.Biol Chem. 2008 Jul;389(7):845-50. doi: 10.1515/BC.2008.098. Biol Chem. 2008. PMID: 18627312 Review.
-
[Interaction between microRNAs and OCT4].Zhongguo Fei Ai Za Zhi. 2015 Jan;18(1):55-8. doi: 10.3779/j.issn.1009-3419.2015.01.09. Zhongguo Fei Ai Za Zhi. 2015. PMID: 25603874 Free PMC article. Review. Chinese.
Cited by
-
MBD6 is a direct target of Oct4 and controls the stemness and differentiation of adipose tissue-derived stem cells.Cell Mol Life Sci. 2013 Feb;70(4):711-28. doi: 10.1007/s00018-012-1157-4. Epub 2012 Sep 30. Cell Mol Life Sci. 2013. PMID: 23052207 Free PMC article.
-
Proteomic profiling of rabbit embryonic stem cells derived from parthenotes and fertilized embryos.PLoS One. 2013 Jul 4;8(7):e67772. doi: 10.1371/journal.pone.0067772. Print 2013. PLoS One. 2013. PMID: 23861804 Free PMC article.
-
Neonatal Death and Heart Failure in Mouse with Transgenic HSP60 Expression.Biomed Res Int. 2015;2015:539805. doi: 10.1155/2015/539805. Epub 2015 Oct 4. Biomed Res Int. 2015. PMID: 26504810 Free PMC article.
-
The Heat Shock Protein HSP70 Promotes Th17 Genes' Expression via Specific Regulation of microRNA.Int J Mol Sci. 2020 Apr 17;21(8):2823. doi: 10.3390/ijms21082823. Int J Mol Sci. 2020. PMID: 32316658 Free PMC article.
-
Extracellular HSP60 triggers tissue regeneration and wound healing by regulating inflammation and cell proliferation.NPJ Regen Med. 2016;1:16013-. doi: 10.1038/npjregenmed.2016.13. Epub 2016 Oct 27. NPJ Regen Med. 2016. PMID: 28936359 Free PMC article.
References
-
- Boyer LA. Plath K. Zeitlinger J. Brambrink T. Medeiros LA. Lee TI. Levine SS. Wernig M. Tajonar A. Ray MK. Bell GA. Otte AP. Vidal M. Gifford DK. Young RA. Jaenisch R. Polycomb complexes developmental regulators in murine embryonic stem cells. Nature. 2006;441:349–353. - PubMed
-
- Bukau B. Horwich AL. The Hsp70 and Hsp60 chaperone machines. Cell. 1998;92:351–366. - PubMed
-
- Bushati N. Cohen SM. microRNA functions. Annu Rev Cell Dev Biol. 2007;23:175–205. - PubMed
-
- Calderwood SK. Mambula SS. Gray PJ., Jr Theriault JR. Extracellular heat shock proteins in cell signaling. FEBS Lett. 2007;581:3689–3694. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

