Interferon-γ release assays for active pulmonary tuberculosis diagnosis in adults in low- and middle-income countries: systematic review and meta-analysis
- PMID: 21996694
- PMCID: PMC3192542
- DOI: 10.1093/infdis/jir410
Interferon-γ release assays for active pulmonary tuberculosis diagnosis in adults in low- and middle-income countries: systematic review and meta-analysis
Abstract
Background: The diagnostic value of interferon-γ release assays (IGRAs) for active tuberculosis in low- and middle-income countries is unclear.
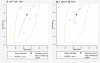
Methods: We searched multiple databases for studies published through May 2010 that evaluated the diagnostic performance of QuantiFERON-TB Gold In-Tube (QFT-GIT) and T-SPOT.TB (T-SPOT) among adults with suspected active pulmonary tuberculosis or patients with confirmed cases in low- and middle-income countries. We summarized test performance characteristics with use of forest plots, hierarchical summary receiver operating characteristic (HSROC) curves, and bivariate random effects models.
Results: Our search identified 789 citations, of which 27 observational studies (17 QFT-GIT and 10 T-SPOT) evaluating 590 human immunodeficiency virus (HIV)-uninfected and 844 HIV-infected individuals met inclusion criteria. Among HIV-infected patients, HSROC/bivariate pooled sensitivity estimates (highest quality data) were 76% (95% confidence interval [CI], 45%-92%) for T-SPOT and 60% (95% CI, 34%-82%) for QFT-GIT. HSROC/bivariate pooled specificity estimates were low for both IGRA platforms among all participants (T-SPOT, 61% [95% CI, 40%-79%]; QFT-GIT, 52% [95% CI, 41%-62%]) and among HIV-infected persons (T-SPOT, 52% [95% CI, 40%-63%]; QFT-GIT, 50% [95% CI, 35%-65%]). There was no consistent evidence that either IGRA was more sensitive than the tuberculin skin test for active tuberculosis diagnosis.
Conclusions: In low- and middle-income countries, neither the tuberculin skin test nor IGRAs have value for active tuberculosis diagnosis in adults, especially in the context of HIV coinfection.
Figures




References
-
- Pai M, Kalantri S, Dheda K. New tools and emerging technologies for the diagnosis of tuberculosis: part I. Latent tuberculosis. Expert Rev Mol Diagn. 2006;6:413–22. - PubMed
-
- Menzies D, Pai M, Comstock G. Meta-analysis: new tests for the diagnosis of latent tuberculosis infection: areas of uncertainty and recommendations for research. Ann Intern Med. 2007;146:340–54. - PubMed
-
- Pai M, Riley LW, Colford JM., Jr Interferon-gamma assays in the immunodiagnosis of tuberculosis: a systematic review. Lancet Infect Dis. 2004;4:761–76. - PubMed
-
- Mazurek G, Jereb J, Vernon A, LoBue P, Goldberg S. Updated guidelines for using interferon-gamma release assays to detect Mycobacterium tuberculosis infection, United States. MMWR. In press. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

