APOL1 localization in normal kidney and nondiabetic kidney disease
- PMID: 21997392
- PMCID: PMC3231786
- DOI: 10.1681/ASN.2011010069
APOL1 localization in normal kidney and nondiabetic kidney disease
Abstract
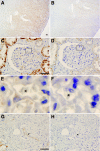
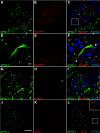
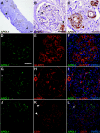
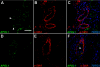
In patients of African ancestry, genetic variants in APOL1, which encodes apolipoprotein L1, associate with the nondiabetic kidney diseases, focal segmental glomerulosclerosis (FSGS), HIV-associated nephropathy (HIVAN), and hypertensive nephropathy. Understanding the renal localization of APOL1 may provide clues that will ultimately help elucidate the mechanisms by which APOL1 variants promote nephropathy. Here, we used immunohistology to examine APOL1 localization in normal human kidney sections and in biopsies demonstrating either FSGS (n = 8) or HIVAN (n = 2). Within normal glomeruli, APOL1 only localized to podocytes. Compared with normal glomeruli, fewer cells stained for APOL1 in FSGS and HIVAN glomeruli, even when expression of the podocyte markers GLEPP1 and synaptopodin appeared normal. APOL1 localized to proximal tubular epithelia in normal kidneys, FSGS, and HIVAN. We detected APOL1 in the arteriolar endothelium of normal and diseased kidney sections. Unexpectedly, in both FSGS and HIVAN but not normal kidneys, the media of medium artery and arterioles contained a subset of α-smooth muscle actin-positive cells that stained for APOL1. Comparing the renal distribution of APOL1 in nondiabetic kidney disease to normal kidney suggests that a previously unrecognized arteriopathy may contribute to disease pathogenesis in patients of African ancestry.
Figures

Comment in
-
Apolipoprotein l1 and the genetic basis for racial disparity in chronic kidney disease.J Am Soc Nephrol. 2011 Nov;22(11):1955-8. doi: 10.1681/ASN.2011090932. Epub 2011 Oct 13. J Am Soc Nephrol. 2011. PMID: 21997393 No abstract available.
References
-
- Kao WH, Klag MJ, Meoni LA, Reich D, Berthier-Schaad Y, Li M, Coresh J, Patterson N, Tandon A, Powe NR, Fink NE, Sadler JH, Weir MR, Abboud HE, Adler SG, Divers J, Iyengar SK, Freedman BI, Kimmel PL, Knowler WC, Kohn OF, Kramp K, Leehey DJ, Nicholas SB, Pahl MV, Schelling JR, Sedor JR, Thornley-Brown D, Winkler CA, Smith MW, Parekh RS: MYH9 is associated with nondiabetic end-stage renal disease in African Americans. Nat Genet 40: 1185–1192, 2008 - PMC - PubMed
-
- Kopp JB, Smith MW, Nelson GW, Johnson RC, Freedman BI, Bowden DW, Oleksyk T, McKenzie LM, Kajiyama H, Ahuja TS, Berns JS, Briggs W, Cho ME, Dart RA, Kimmel PL, Korbet SM, Michel DM, Mokrzycki MH, Schelling JR, Simon E, Trachtman H, Vlahov D, Winkler CA: MYH9 is a major-effect risk gene for focal segmental glomerulosclerosis. Nat Genet 40: 1175–1184, 2008 - PMC - PubMed
-
- Arrondel C, Vodovar N, Knebelmann B, Grunfeld JP, Gubler MC, Antignac C, Heidet L: Expression of the nonmuscle myosin heavy chain IIA in the human kidney and screening for MYH9 mutations in Epstein and Fechtner syndromes. J Am Soc Nephrol 13: 65–74, 2002 - PubMed
-
- Ghiggeri GM, Caridi G, Magrini U, Sessa A, Savoia A, Seri M, Pecci A, Romagnoli R, Gangarossa S, Noris P, Sartore S, Necchi V, Ravazzolo R, Balduini CL: Genetics, clinical and pathological features of glomerulonephritis associated with mutations of nonmuscle myosin IIA (Fechtner syndrome). Am J Kidney Dis 41: 95–104, 2003 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

