A potent and broad neutralizing antibody recognizes and penetrates the HIV glycan shield
- PMID: 21998254
- PMCID: PMC3280215
- DOI: 10.1126/science.1213256
A potent and broad neutralizing antibody recognizes and penetrates the HIV glycan shield
Abstract
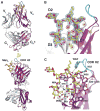
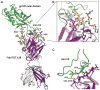
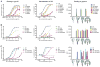
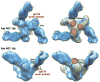
The HIV envelope (Env) protein gp120 is protected from antibody recognition by a dense glycan shield. However, several of the recently identified PGT broadly neutralizing antibodies appear to interact directly with the HIV glycan coat. Crystal structures of antigen-binding fragments (Fabs) PGT 127 and 128 with Man(9) at 1.65 and 1.29 angstrom resolution, respectively, and glycan binding data delineate a specific high mannose-binding site. Fab PGT 128 complexed with a fully glycosylated gp120 outer domain at 3.25 angstroms reveals that the antibody penetrates the glycan shield and recognizes two conserved glycans as well as a short β-strand segment of the gp120 V3 loop, accounting for its high binding affinity and broad specificity. Furthermore, our data suggest that the high neutralization potency of PGT 127 and 128 immunoglobulin Gs may be mediated by cross-linking Env trimers on the viral surface.
Figures

References
-
- Wyatt R, Sodroski J. The HIV-1 envelope glycoproteins: fusogens, antigens, and immunogens. Science. 1998;280:1884–1888. - PubMed
-
- Toone EJ. Structure and energetics of protein-carbohydrate complexes. Curr Opin Struct Biol. 1994;4:719–728.
-
- Calarese DA, et al. Antibody domain exchange is an immunological solution to carbohydrate cluster recognition. Science. 2003;300:2065–2071. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
Grants and funding
- Y1-GM-1104/GM/NIGMS NIH HHS/United States
- P41 RR017573/RR/NCRR NIH HHS/United States
- P41 RR001209/RR/NCRR NIH HHS/United States
- AI33292/AI/NIAID NIH HHS/United States
- P01 AI082362/AI/NIAID NIH HHS/United States
- F32 AI074372/AI/NIAID NIH HHS/United States
- RR017573/RR/NCRR NIH HHS/United States
- P41RR001209/RR/NCRR NIH HHS/United States
- R01 AI084817/AI/NIAID NIH HHS/United States
- HFE-224662/CAPMC/ CIHR/Canada
- R56 AI084817/AI/NIAID NIH HHS/United States
- AI84817/AI/NIAID NIH HHS/United States
- R37 AI033292/AI/NIAID NIH HHS/United States
- AI74372/AI/NIAID NIH HHS/United States
- R01 AI033292/AI/NIAID NIH HHS/United States
- Y1-CO-1020/CO/NCI NIH HHS/United States
- AI082362/AI/NIAID NIH HHS/United States
- U01 CA128416/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources

