Exogenous addition of a C-xylopyranoside derivative stimulates keratinocyte dermatan sulfate synthesis and promotes migration
- PMID: 21998662
- PMCID: PMC3187761
- DOI: 10.1371/journal.pone.0025480
Exogenous addition of a C-xylopyranoside derivative stimulates keratinocyte dermatan sulfate synthesis and promotes migration
Abstract
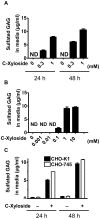
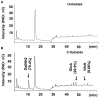
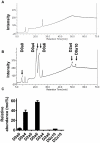
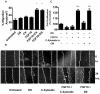
As C-Xyloside has been suggested to be an initiator of glycosaminoglycan (GAG) synthesis, and GAGs such as Dermatan sulfate (DS) are potent enhancers of fibroblast growth factor (FGF)--10 action, we investigated if a C-Xylopyranoside derivative, (C-β-D-xylopyranoside-2-hydroxy-propane, C-Xyloside), could promote DS production by cultured normal human keratinocytes, how this occurs and if C-Xyloside could also stimulate FGF-dependent cell migration and proliferation. C-Xyloside-treated keratinocytes greatly increased secretion of total sulfated GAGs. Majority of the induced GAG was chondroitin sulfate/dermatan sulfate (CS/DS) of which the major secreted GAG was DS. Cells lacking xylosyltransferase enzymatic activity demonstrated that C-Xyloside was able to stimulate GAG synthesis without addition to core proteins. Consistent with the observed increase in DS, keratinocytes treated with C-Xyloside showed enhanced migration in response to FGF-10 and secreted into their culture media GAGs that promoted FGF-10-dependent cellular proliferation. These results indicate that C-Xyloside may enhance epithelial repair by serving as an initiator of DS synthesis.
Conflict of interest statement
Figures
References
-
- Penc SF, Pomahac B, Winkler T, Dorschner RA, Eriksson E, et al. Dermatan sulfate released after injury is a potent promoter of fibroblast growth factor-2 function. J Biol Chem. 1998;273:28116–28121. - PubMed
-
- Taylor KR, Rudisill JA, Gallo RL. Structural and sequence motifs in dermatan sulfate for promoting fibroblast growth factor-2 (FGF-2) and FGF-7 activity. J Biol Chem. 2005;280:5300–5306. - PubMed
-
- Ashikari S, Habuchi H, Kimata K. Characterization of heparan sulfate oligosaccharides that bind to hepatocyte growth factor. J Biol Chem. 1995;270:29586–29593. - PubMed
-
- Lyon M, Deakin JA, Rahmoune H, Fernig DG, Nakamura T, et al. Hepatocyte growth factor/scatter factor binds with high affinity to dermatan sulfate. J Biol Chem. 1998;273:271–278. - PubMed
-
- Maccarana M, Casu B, Lindahl U. Minimal sequence in heparin/heparan sulfate required for binding of basic fibroblast growth factor. J Biol Chem. 1993;268:23898–23905. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

