Membrane-bound steel factor maintains a high local concentration for mouse primordial germ cell motility, and defines the region of their migration
- PMID: 21998739
- PMCID: PMC3188585
- DOI: 10.1371/journal.pone.0025984
Membrane-bound steel factor maintains a high local concentration for mouse primordial germ cell motility, and defines the region of their migration
Abstract
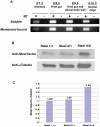
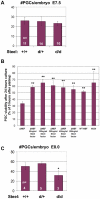
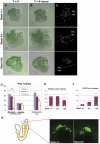
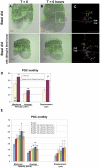
Steel factor, the protein product of the Steel locus in the mouse, is a multifunctional signal for the primordial germ cell population. We have shown previously that its expression accompanies the germ cells during migration to the gonads, forming a "travelling niche" that controls their survival, motility, and proliferation. Here we show that these functions are distributed between the alternatively spliced membrane-bound and soluble forms of Steel factor. The germ cells normally migrate as individuals from E7.5 to E11.5, when they aggregate together in the embryonic gonads. Movie analysis of Steel-dickie mutant embryos, which make only the soluble form, at E7.5, showed that the germ cells fail to migrate normally, and undergo "premature aggregation" in the base of the allantois. Survival and directionality of movement is not affected. Addition of excess soluble Steel factor to Steel-dickie embryos rescued germ cell motility, and addition of Steel factor to germ cells in vitro showed that a fourfold higher dose was required to increase motility, compared to survival. These data show that soluble Steel factor is sufficient for germ cell survival, and suggest that the membrane-bound form provides a higher local concentration of Steel factor that controls the balance between germ cell motility and aggregation. This hypothesis was tested by addition of excess soluble Steel factor to slice cultures of E11.5 embryos, when migration usually ceases, and the germ cells aggregate. This reversed the aggregation process, and caused increased motility of the germ cells. We conclude that the two forms of Steel factor control different aspects of germ cell behavior, and that membrane-bound Steel factor controls germ cell motility within a "motility niche" that moves through the embryo with the germ cells. Escape from this niche causes cessation of motility and death by apoptosis of the ectopic germ cells.
Conflict of interest statement
Figures

References
-
- Hayashi K, de Sousa Lopes SM, Surani MA. Germ cell specification in mice. Science. 2007;316:394–396. - PubMed
-
- Ohinata Y, Payer B, O'Carroll D, Ancelin K, Ono Y, et al. Blimp1 is a critical determinant of the germ cell lineage in mice. Nature. 2005;436:207–213. - PubMed
-
- Anderson R, Copeland TK, Scholer H, Heasman J, Wylie C. The onset of germ cell migration in the mouse embryo. Mech Dev. 2000;91:61–68. - PubMed
-
- Gu Y, Runyan C, Shoemaker A, Surani A, Wylie C. Steel factor controls primordial germ cell survival and motility from the time of their specification in the allantois, and provides a continuous niche throughout their migration. Development. 2009;136:1295–1303. - PubMed
-
- Molyneaux KA, Stallock J, Schaible K, Wylie C. Time-lapse analysis of living mouse germ cell migration. Dev Biol. 2001;240:488–498. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

