Coding-independent regulation of the tumor suppressor PTEN by competing endogenous mRNAs
- PMID: 22000013
- PMCID: PMC3235920
- DOI: 10.1016/j.cell.2011.09.029
Coding-independent regulation of the tumor suppressor PTEN by competing endogenous mRNAs
Abstract
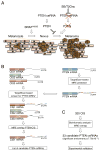
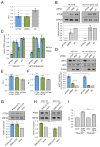
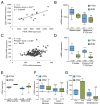
Here, we demonstrate that protein-coding RNA transcripts can crosstalk by competing for common microRNAs, with microRNA response elements as the foundation of this interaction. We have termed such RNA transcripts as competing endogenous RNAs (ceRNAs). We tested this hypothesis in the context of PTEN, a key tumor suppressor whose abundance determines critical outcomes in tumorigenesis. By a combined computational and experimental approach, we identified and validated endogenous protein-coding transcripts that regulate PTEN, antagonize PI3K/AKT signaling, and possess growth- and tumor-suppressive properties. Notably, we also show that these genes display concordant expression patterns with PTEN and copy number loss in cancers. Our study presents a road map for the prediction and validation of ceRNA activity and networks and thus imparts a trans-regulatory function to protein-coding mRNAs.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures




Comment in
-
Epigenetics. Layer by layer.Nat Rev Cancer. 2011 Nov 3;11(12):830. doi: 10.1038/nrc3172. Nat Rev Cancer. 2011. PMID: 22048565 No abstract available.
-
Regulatory RNA: layer by layer.Nat Rev Genet. 2011 Nov 3;12(12):804. doi: 10.1038/nrg3108. Nat Rev Genet. 2011. PMID: 22048663 No abstract available.
-
RNA: a new layer of regulation.Nat Rev Mol Cell Biol. 2011 Nov 3;12(12):766. doi: 10.1038/nrm3225. Nat Rev Mol Cell Biol. 2011. PMID: 22048709 No abstract available.
-
ceRNAs: miRNA target mimic mimics.Cell. 2011 Dec 23;147(7):1431-2. doi: 10.1016/j.cell.2011.12.003. Cell. 2011. PMID: 22196719 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

