Enhanced growth of Acidovorax sp. strain 2AN during nitrate-dependent Fe(II) oxidation in batch and continuous-flow systems
- PMID: 22003007
- PMCID: PMC3233087
- DOI: 10.1128/AEM.06214-11
Enhanced growth of Acidovorax sp. strain 2AN during nitrate-dependent Fe(II) oxidation in batch and continuous-flow systems
Abstract
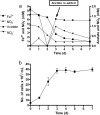
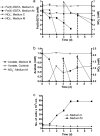
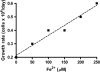
Microbial nitrate-dependent, Fe(II) oxidation (NDFO) is a ubiquitous biogeochemical process in anoxic sediments. Since most microorganisms that can oxidize Fe(II) with nitrate require an additional organic substrate for growth or sustained Fe(II) oxidation, the energetic benefits of NDFO are unclear. The process may also be self-limiting in batch cultures due to formation of Fe-oxide cell encrustations. We hypothesized that NDFO provides energetic benefits via a mixotrophic physiology in environments where cells encounter very low substrate concentrations, thereby minimizing cell encrustations. Acidovorax sp. strain 2AN was incubated in anoxic batch reactors in a defined medium containing 5 to 6 mM NO₃⁻, 8 to 9 mM Fe²⁺, and 1.5 mM acetate. Almost 90% of the Fe(II) was oxidized within 7 days with concomitant reduction of nitrate and complete consumption of acetate. Batch-grown cells became heavily encrusted with Fe(III) oxyhydroxides, lost motility, and formed aggregates. Encrusted cells could neither oxidize more Fe(II) nor utilize further acetate additions. In similar experiments with chelated iron (Fe(II)-EDTA), encrusted cells were not produced, and further additions of acetate and Fe(II)-EDTA could be oxidized. Experiments using a novel, continuous-flow culture system with low concentrations of substrate, e.g., 100 μM NO₃⁻, 20 μM acetate, and 50 to 250 μM Fe²⁺, showed that the growth yield of Acidovorax sp. strain 2AN was always greater in the presence of Fe(II) than in its absence, and electron microscopy showed that encrustation was minimized. Our results provide evidence that, under environmentally relevant concentrations of substrates, NDFO can enhance growth without the formation of growth-limiting cell encrustations.
Figures
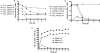


Similar articles
-
Induction of nitrate-dependent Fe(II) oxidation by Fe(II) in Dechloromonas sp. strain UWNR4 and Acidovorax sp. strain 2AN.Appl Environ Microbiol. 2013 Jan;79(2):748-52. doi: 10.1128/AEM.02709-12. Epub 2012 Nov 9. Appl Environ Microbiol. 2013. PMID: 23144134 Free PMC article.
-
Oxidation of Fe(II)-EDTA by nitrite and by two nitrate-reducing Fe(II)-oxidizing Acidovorax strains.Geobiology. 2015 Mar;13(2):198-207. doi: 10.1111/gbi.12125. Epub 2015 Jan 22. Geobiology. 2015. PMID: 25612223
-
Potential role of nitrite for abiotic Fe(II) oxidation and cell encrustation during nitrate reduction by denitrifying bacteria.Appl Environ Microbiol. 2014 Feb;80(3):1051-61. doi: 10.1128/AEM.03277-13. Epub 2013 Nov 22. Appl Environ Microbiol. 2014. PMID: 24271182 Free PMC article.
-
Autotrophic denitrification using Fe(II) as an electron donor: A novel prospective denitrification process.Sci Total Environ. 2023 Feb 1;858(Pt 1):159721. doi: 10.1016/j.scitotenv.2022.159721. Epub 2022 Oct 25. Sci Total Environ. 2023. PMID: 36306837 Review.
-
Microbially mediated coupling of nitrate reduction and Fe(II) oxidation under anoxic conditions.FEMS Microbiol Ecol. 2019 Apr 1;95(4):fiz030. doi: 10.1093/femsec/fiz030. FEMS Microbiol Ecol. 2019. PMID: 30844067 Review.
Cited by
-
Sphaerotilus natans encrusted with nanoball-shaped Fe(III) oxide minerals formed by nitrate-reducing mixotrophic Fe(II) oxidation.FEMS Microbiol Ecol. 2014 Oct;90(1):68-77. doi: 10.1111/1574-6941.12372. Epub 2014 Jul 9. FEMS Microbiol Ecol. 2014. PMID: 24965827 Free PMC article.
-
Neutrophilic, nitrate-dependent, Fe(II) oxidation by a Dechloromonas species.World J Microbiol Biotechnol. 2013 Apr;29(4):617-23. doi: 10.1007/s11274-012-1217-9. Epub 2012 Nov 27. World J Microbiol Biotechnol. 2013. PMID: 23184578
-
Underestimation about the Contribution of Nitrate Reducers to Iron Cycling Indicated by Enterobacter Strain.Molecules. 2022 Aug 30;27(17):5581. doi: 10.3390/molecules27175581. Molecules. 2022. PMID: 36080348 Free PMC article.
-
Salinity Impact on Composition and Activity of Nitrate-Reducing Fe(II)-Oxidizing Microorganisms in Saline Lakes.Appl Environ Microbiol. 2022 May 24;88(10):e0013222. doi: 10.1128/aem.00132-22. Epub 2022 May 2. Appl Environ Microbiol. 2022. PMID: 35499328 Free PMC article.
-
Ligand-enhanced abiotic iron oxidation and the effects of chemical versus biological iron cycling in anoxic environments.Environ Sci Technol. 2013 Mar 19;47(6):2602-11. doi: 10.1021/es3049459. Epub 2013 Feb 27. Environ Sci Technol. 2013. PMID: 23402562 Free PMC article.
References
-
- Atlas R. M. 2004. Handbook of media for environmental microbiology, 3rd ed CRC Press, Inc., Boca Raton, FL
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

