Trigeminal ganglion neuron subtype-specific alterations of Ca(V)2.1 calcium current and excitability in a Cacna1a mouse model of migraine
- PMID: 22005682
- PMCID: PMC3249056
- DOI: 10.1113/jphysiol.2011.220533
Trigeminal ganglion neuron subtype-specific alterations of Ca(V)2.1 calcium current and excitability in a Cacna1a mouse model of migraine
Abstract
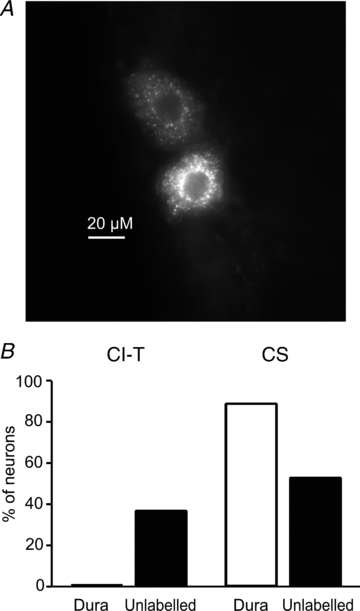
Familial hemiplegic migraine type-1 (FHM1), a monogenic subtype of migraine with aura, is caused by gain-of-function mutations in Ca(V)2.1 (P/Q-type) calcium channels. The consequences of FHM1 mutations on the trigeminovascular pathway that generates migraine headache remain largely unexplored. Here we studied the calcium currents and excitability properties of two subpopulations of small-diameter trigeminal ganglion (TG) neurons from adult wild-type (WT) and R192Q FHM1 knockin (KI) mice: capsaicin-sensitive neurons without T-type calcium currents (CS) and capsaicin-insensitive neurons characterized by the expression of T-type calcium currents (CI-T). Small TG neurons retrogradely labelled from the dura are mostly CS neurons, while CI-T neurons were not present in the labelled population. CS and CI-T neurons express Ca(V)2.1 channels with different activation properties, and the Ca(V)2.1 channels are differently affected by the FHM1 mutation in the two TG neuron subtypes. In CI-T neurons from FHM1 KI mice there was a larger P/Q-type current density following mild depolarizations, a larger action potential (AP)-evoked calcium current and a longer AP duration when compared to CI-T neurons from WT mice. In striking contrast, the P/Q-type current density, voltage dependence and kinetics were not altered by the FHM1 mutation in CS neurons. The excitability properties of mutant CS neurons were also unaltered. Congruently, the FHM1 mutation did not alter depolarization-evoked CGRP release from the dura mater, while CGRP release from the trigeminal ganglion was larger in KI compared to WT mice. Our findings suggest that the facilitation of peripheral mechanisms of CGRP action, such as dural vasodilatation and nociceptor sensitization at the meninges, does not contribute to the generation of headache in FHM1.
Figures



Comment in
-
Unequal gains of function are a headache for migraine mechanisms.J Physiol. 2012 Jan 1;590(1):1-2. doi: 10.1113/jphysiol.2011.223545. J Physiol. 2012. PMID: 22210282 Free PMC article. No abstract available.
References
-
- Adams PJ, Garcia E, David LS, Mulatz KJ, Spacey SD, Snutch TP. CaV2.1 P/Q-type calcium channel alternative splicing affects the functional impact of familial hemiplegic migraine mutations: implications for calcium channelopathies. Channels (Austin) 2009;3:110–121. - PubMed
-
- Ayata C. Spreading depression: from serendipity to targeted therapy in migraine prophylaxis. Cephalalgia. 2009;29:1095–1114. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

