Taking STEPs forward to understand fragile X syndrome
- PMID: 22009355
- PMCID: PMC3922766
- DOI: 10.1007/978-3-642-21649-7_12
Taking STEPs forward to understand fragile X syndrome
Abstract
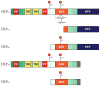
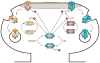
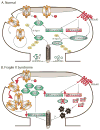
A priority of fragile X syndrome (FXS) research is to determine the molecular mechanisms underlying the functional, behavioral, and structural deficits in humans and in the FXS mouse model. Given that metabotropic glutamate receptor (mGluR) long-term depression (LTD) is exaggerated in FXS mice, considerable effort has focused on proteins that regulate this form of synaptic plasticity. STriatal-Enriched protein tyrosine Phosphatase (STEP) is a brain-specific phosphatase implicated as an "LTD protein" because it mediates AMPA receptor internalization during mGluR LTD. STEP also promotes NMDA receptor endocytosis and inactivates ERK1/2 and Fyn, thereby opposing synaptic strengthening. We hypothesized that dysregulation of STEP may contribute to the pathophysiology of FXS. We review how STEP's expression and activity are regulated by dendritic protein synthesis, ubiquitination, proteolysis, and phosphorylation. We also discuss implications for STEP in FXS and other disorders, including Alzheimer's disease. As highlighted here, pharmacological interventions targeting STEP may prove successful for FXS.
Figures
References
-
- Bear MF, Huber KM, Warren ST. The mGluR theory of fragile X mental retardation. Trends Neurosci. 2004;27:370–377. - PubMed
-
- Besshoh S, Bawa D, Teves L, Wallace MC, Gurd JW. Increased phosphorylation and redistribution of NMDA receptors between synaptic lipid rafts and post-synaptic densities following transient global ischemia in the rat brain. J Neurochem. 2005;93:186–194. - PubMed
-
- Blanpied TA, Scott DB, Ehlers MD. Dynamics and regulation of clathrin coats at specialized endocytic zones of dendrites and spines. Neuron. 2002;36:435–449. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

