Endogenous annexin A1 counter-regulates bleomycin-induced lung fibrosis
- PMID: 22011168
- PMCID: PMC3212807
- DOI: 10.1186/1471-2172-12-59
Endogenous annexin A1 counter-regulates bleomycin-induced lung fibrosis
Abstract
Background: The balancing functions of pro/anti-inflammatory mediators of the complex innate responses have been investigated in a variety of experimental inflammatory settings. Annexin-A1 (AnxA1) is one mediator of endogenous anti-inflammation, affording regulation of leukocyte trafficking and activation in many contexts, yet its role in lung pathologies has been scarcely investigated, despite being highly expressed in lung cells. Here we have applied the bleomycin lung fibrosis model to AnxA1 null mice over a 21-day time-course, to monitor potential impact of this mediator on the control of the inflammatory and fibrotic phases.
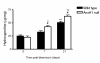
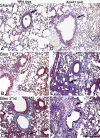
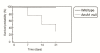
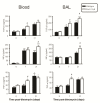
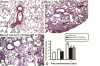
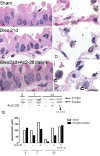
Results: Analyses in wild-type mice revealed strict spatial and temporal regulation of the Anxa1 gene, e.g. up-regulation in epithelial cells and infiltrated granulocytes at day 7, followed by augmented protein levels in alveolar macrophages by day 21. Absence of AnxA1 caused increases in: i) the degree of inflammation at day 7; and ii) indexes of fibrosis (assessed by deposition of hydroxyproline in the lung) at day 7 and 21. These alterations in AnxA1 null mice were paralleled by augmented TGF-β1, IFN-γ and TNF-α generation compared to wild-type mice. Finally, treatment of wild type animals with an AnxA1 peptido-mimetic, given prophylactically (from day 0 to 21) or therapeutically (from day 14 onward), ameliorated both signs of inflammation and fibrosis.
Conclusion: Collectively these data reveal a pathophysiological relevance for endogenous AnxA1 in lung inflammation and, more importantly, fibrosis, and may open new insights for the pharmacological treatment of lung fibrosis.
Figures

References
-
- Moore BB, Hogaboam CM. Murine models of pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol. 2008;294:L152–L160. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

