Fructose-1,6-bisphosphate aldolase (class II) is the primary site of nickel toxicity in Escherichia coli
- PMID: 22014167
- PMCID: PMC3225726
- DOI: 10.1111/j.1365-2958.2011.07891.x
Fructose-1,6-bisphosphate aldolase (class II) is the primary site of nickel toxicity in Escherichia coli
Abstract
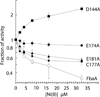
Nickel is toxic to all forms of life, but the mechanisms of cell damage are unknown. Indeed, environmentally relevant nickel levels (8 µM) inhibit wild-type Escherichia coli growth on glucose minimal medium. The same concentration of nickel also inhibits growth on fructose, but not succinate, lactate or glycerol; these results suggest that fructose-1,6-bisphosphate aldolase (FbaA) is a target of nickel toxicity. Cells stressed by 8 µM Ni(II) for 20 min lost 75% of their FbaA activity, demonstrating that FbaA is inactivated during nickel stress. Furthermore, overexpression of fbaA restored growth of an rcnA mutant in glucose minimal medium supplemented with 4 µM Ni(II), thus confirming that FbaA is a primary target of nickel toxicity. This class II aldolase has an active site zinc and a non-catalytic zinc nearby. Purified FbaA lost 80 % of its activity within 2 min when challenged with 8 µM Ni(II). Nickel-challenged FbaA lost 0.8 zinc and gained 0.8 nickel per inactivated monomer. FbaA mutants (D144A and E174A) affecting the non-catalytic zinc were resistant to nickel inhibition. These results define the primary site of nickel toxicity in E. coli as the class II aldolase FbaA through binding to the non-catalytic zinc site.
© 2011 Blackwell Publishing Ltd.
Figures






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

