Multiple adaptable mechanisms early in the primate visual pathway
- PMID: 22016535
- PMCID: PMC3271438
- DOI: 10.1523/JNEUROSCI.0890-11.2011
Multiple adaptable mechanisms early in the primate visual pathway
Abstract
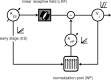
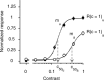
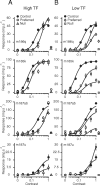
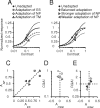
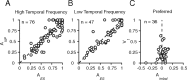
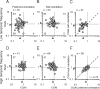
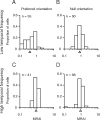
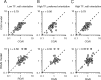
We describe experiments that isolate and characterize multiple adaptable mechanisms that influence responses of orientation-selective neurons in primary visual cortex (V1) of anesthetized macaque (Macaca fascicularis). The results suggest that three adaptable stages of machinery shape neural responses in V1: a broadly tuned early stage and a spatio-temporally tuned later stage, both of which provide excitatory input, and a normalization pool that is also broadly tuned. The early stage and the normalization pool are revealed by adapting gratings that themselves fail to evoke a response from the neuron: either low temporal frequency gratings at the null orientation or gratings of any orientation drifting at high temporal frequencies. When effective, adapting stimuli that altered the sensitivity of these two mechanisms caused reductions of contrast gain and often brought about a paradoxical increase in response gain due to a relatively greater desensitization of the normalization pool. The tuned mechanism is desensitized only by stimuli well matched to a neuron's receptive field. We could thus infer desensitization of the tuned mechanism by comparing effects obtained with adapting gratings of preferred and null orientation modulated at low temporal frequencies.
Figures
References
-
- Albrecht DG, Hamilton DB. Striate cortex of monkey and cat: contrast response function. J Neurophysiol. 1982;48:217–237. - PubMed
-
- Allison JD, Melzer P, Ding Y, Bonds AB, Casagrande VA. Differential contributions of magnocellular and parvocellular pathways to the contrast response of neurons in bush baby primary visual cortex (V1) Vis Neurosci. 2000;17:71–76. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
