Dissociation of dorsal hippocampal regional activation under the influence of stress in freely behaving rats
- PMID: 22022311
- PMCID: PMC3194099
- DOI: 10.3389/fnbeh.2011.00066
Dissociation of dorsal hippocampal regional activation under the influence of stress in freely behaving rats
Abstract
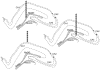
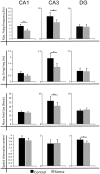
Stress has deleterious effects on brain, body, and behavior in humans and animals alike. The present work investigated how 30-min acute photic stress exposure impacts on spatial information processing in the main sub-regions of the dorsal hippocampal formation [CA1, CA3, and dentate gyrus (DG)], a brain structure prominently implicated in memory and spatial representation. Recordings were performed from spatially tuned hippocampal and DG cells in rats while animals foraged in a square arena for food. The stress procedure induced a decrease in firing frequencies in CA1 and CA3 place cells while sparing locational characteristics. In contrast to the CA1-CA3 network, acute stress failed to induce major changes in the DG neuronal population. These data demonstrate a clear dissociation of the effects of stress on the main hippocampal sub-regions. Our findings further support the notion of decreased hippocampal excitability arising from behavioral stress in areas CA1 and CA3, but not in DG.
Keywords: CA1; CA3; dentate gyrus; freely moving; hippocampus; place cells; stress.
Figures
Similar articles
-
Input-output relations in the entorhinal cortex-dentate-hippocampal system: evidence for a non-linear transfer of signals.Neuroscience. 2006 Sep 29;142(1):247-65. doi: 10.1016/j.neuroscience.2006.06.001. Epub 2006 Jul 14. Neuroscience. 2006. PMID: 16844310
-
Ensemble place codes in hippocampus: CA1, CA3, and dentate gyrus place cells have multiple place fields in large environments.PLoS One. 2011;6(7):e22349. doi: 10.1371/journal.pone.0022349. Epub 2011 Jul 15. PLoS One. 2011. PMID: 21789250 Free PMC article.
-
Dentate Gyrus Sharp Waves, a Local Field Potential Correlate of Learning in the Dentate Gyrus of Mice.J Neurosci. 2020 Sep 9;40(37):7105-7118. doi: 10.1523/JNEUROSCI.2275-19.2020. Epub 2020 Aug 19. J Neurosci. 2020. PMID: 32817247 Free PMC article.
-
Tracking the flow of hippocampal computation: Pattern separation, pattern completion, and attractor dynamics.Neurobiol Learn Mem. 2016 Mar;129:38-49. doi: 10.1016/j.nlm.2015.10.008. Epub 2015 Oct 26. Neurobiol Learn Mem. 2016. PMID: 26514299 Free PMC article. Review.
-
Stress modulation of hippocampal activity--spotlight on the dentate gyrus.Neurobiol Learn Mem. 2014 Jul;112:53-60. doi: 10.1016/j.nlm.2014.04.008. Epub 2014 Apr 18. Neurobiol Learn Mem. 2014. PMID: 24747273 Review.
Cited by
-
In vivo Calcium Imaging Reveals That Cortisol Treatment Reduces the Number of Place Cells in Thy1-GCaMP6f Transgenic Mice.Front Neurosci. 2019 Mar 1;13:176. doi: 10.3389/fnins.2019.00176. eCollection 2019. Front Neurosci. 2019. PMID: 30881283 Free PMC article.
-
Chronic Stress Alters Spatial Representation and Bursting Patterns of Place Cells in Behaving Mice.Sci Rep. 2015 Nov 9;5:16235. doi: 10.1038/srep16235. Sci Rep. 2015. PMID: 26548337 Free PMC article.
-
The effects of oxytocin on cognitive defect caused by chronic restraint stress applied to adolescent rats and on hippocampal VEGF and BDNF levels.Med Sci Monit. 2015 Jan 6;21:69-75. doi: 10.12659/MSM.893159. Med Sci Monit. 2015. PMID: 25559382 Free PMC article.
-
Acute stress and hippocampal output: exploring dorsal CA1 and subicular synaptic plasticity simultaneously in anesthetized rats.Physiol Rep. 2013 Jul;1(2):e00035. doi: 10.1002/phy2.35. Epub 2013 Jul 21. Physiol Rep. 2013. PMID: 24303119 Free PMC article.
-
Stress enhances hippocampal neuronal synchrony and alters ripple-spike interaction.Neurobiol Stress. 2021 Apr 13;14:100327. doi: 10.1016/j.ynstr.2021.100327. eCollection 2021 May. Neurobiol Stress. 2021. PMID: 33937446 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

