A naturally occurring C-terminal fragment of the prion protein (PrP) delays disease and acts as a dominant-negative inhibitor of PrPSc formation
- PMID: 22025612
- PMCID: PMC3243553
- DOI: 10.1074/jbc.M111.286195
A naturally occurring C-terminal fragment of the prion protein (PrP) delays disease and acts as a dominant-negative inhibitor of PrPSc formation
Abstract
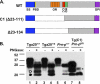
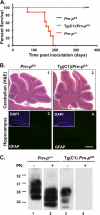
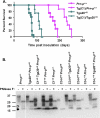
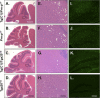
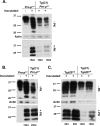
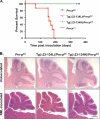
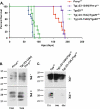
The cellular prion protein (PrPC) undergoes constitutive proteolytic cleavage between residues 111/112 to yield a soluble N-terminal fragment (N1) and a membrane-anchored C-terminal fragment (C1). The C1 fragment represents the major proteolytic fragment of PrPC in brain and several cell types. To explore the role of C1 in prion disease, we generated Tg(C1) transgenic mice expressing this fragment (PrP(Δ23-111)) in the presence and absence of endogenous PrP. In contrast to several other N-terminally deleted forms of PrP, the C1 fragment does not cause a spontaneous neurological disease in the absence of endogenous PrP. Tg(C1) mice inoculated with scrapie prions remain healthy and do not accumulate protease-resistant PrP, demonstrating that C1 is not a substrate for conversion to PrPSc (the disease-associated isoform). Interestingly, Tg(C1) mice co-expressing C1 along with wild-type PrP (either endogenous or encoded by a second transgene) become ill after scrapie inoculation, but with a dramatically delayed time course compared with mice lacking C1. In addition, accumulation of PrPSc was markedly slowed in these animals. Similar effects were produced by a shorter C-terminal fragment of PrP(Δ23-134). These results demonstrate that C1 acts as dominant-negative inhibitor of PrPSc formation and accumulation of neurotoxic forms of PrP. Thus, C1, a naturally occurring fragment of PrPC, might play a modulatory role during the course of prion diseases. In addition, enhancing production of C1, or exogenously administering this fragment, represents a potential therapeutic strategy for the treatment of prion diseases.
Figures

Similar articles
-
Strain-Dependent Prion Infection in Mice Expressing Prion Protein with Deletion of Central Residues 91-106.Int J Mol Sci. 2020 Oct 1;21(19):7260. doi: 10.3390/ijms21197260. Int J Mol Sci. 2020. PMID: 33019549 Free PMC article.
-
Sheep scrapie susceptibility-linked polymorphisms do not modulate the initial binding of cellular to disease-associated prion protein prior to conversion.J Gen Virol. 2005 Sep;86(Pt 9):2627-2634. doi: 10.1099/vir.0.80901-0. J Gen Virol. 2005. PMID: 16099922
-
Dominant-negative inhibition of prion replication in transgenic mice.Proc Natl Acad Sci U S A. 2002 Oct 1;99(20):13079-84. doi: 10.1073/pnas.182425299. Epub 2002 Sep 23. Proc Natl Acad Sci U S A. 2002. PMID: 12271119 Free PMC article.
-
Genetic and infectious prion diseases.Arch Neurol. 1993 Nov;50(11):1129-53. doi: 10.1001/archneur.1993.00540110011002. Arch Neurol. 1993. PMID: 8105771 Review.
-
Prion encephalopathies of animals and humans.Dev Biol Stand. 1993;80:31-44. Dev Biol Stand. 1993. PMID: 8270114 Review.
Cited by
-
Separate mechanisms act concurrently to shed and release the prion protein from the cell.Prion. 2012 Nov-Dec;6(5):498-509. doi: 10.4161/pri.22588. Epub 2012 Oct 23. Prion. 2012. PMID: 23093798 Free PMC article.
-
Neuroprotective effect and potential of cellular prion protein and its cleavage products for treatment of neurodegenerative disorders part I. a literature review.Expert Rev Neurother. 2021 Sep;21(9):969-982. doi: 10.1080/14737175.2021.1965881. Epub 2021 Sep 2. Expert Rev Neurother. 2021. PMID: 34470561 Free PMC article. Review.
-
The Cellular Prion Protein-ROCK Connection: Contribution to Neuronal Homeostasis and Neurodegenerative Diseases.Front Cell Neurosci. 2021 Apr 12;15:660683. doi: 10.3389/fncel.2021.660683. eCollection 2021. Front Cell Neurosci. 2021. PMID: 33912016 Free PMC article. Review.
-
Cellular prion protein regulates its own α-cleavage through ADAM8 in skeletal muscle.J Biol Chem. 2012 May 11;287(20):16510-20. doi: 10.1074/jbc.M112.360891. Epub 2012 Mar 23. J Biol Chem. 2012. PMID: 22447932 Free PMC article.
-
Octarepeat region flexibility impacts prion function, endoproteolysis and disease manifestation.EMBO Mol Med. 2015 Mar;7(3):339-56. doi: 10.15252/emmm.201404588. EMBO Mol Med. 2015. PMID: 25661904 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

