Self-maintained escort cells form a germline stem cell differentiation niche
- PMID: 22031542
- PMCID: PMC3210492
- DOI: 10.1242/dev.067850
Self-maintained escort cells form a germline stem cell differentiation niche
Abstract
Stem cell self-renewal is controlled by concerted actions of niche signals and intrinsic factors in a variety of systems. In the Drosophila ovary, germline stem cells (GSCs) in the niche continuously self-renew and generate differentiated germ cells that interact physically with escort cells (ECs). It has been proposed that escort stem cells (ESCs), which directly contact GSCs, generate differentiated ECs to maintain the EC population. However, it remains unclear whether the differentiation status of germ cells affects EC behavior and how the interaction between ECs and germ cells is regulated. In this study, we have found that ECs can undergo slow cell turnover regardless of their positions, and the lost cells are replenished by their neighboring ECs via self-duplication rather than via stem cells. ECs extend elaborate cellular processes that exhibit extensive interactions with differentiated germ cells. Interestingly, long cellular processes of ECs are absent when GSC progeny fail to differentiate, suggesting that differentiated germ cells are required for the formation or maintenance of EC cellular processes. Disruption of Rho functions leads to the disruption of long EC cellular processes and the accumulation of ill-differentiated single germ cells by increasing BMP signaling activity outside the GSC niche, and also causes gradual EC loss. Therefore, our findings indicate that ECs interact extensively with differentiated germ cells through their elaborate cellular processes and control proper germ cell differentiation. Here, we propose that ECs form a niche that controls GSC lineage differentiation and is maintained by a non-stem cell mechanism.
Figures



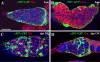
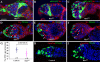


References
-
- Belenkaya T. Y., Han C., Yan D., Opoka R. J., Khodoun M., Liu H., Lin X. (2004). Drosophila Dpp morphogen movement is independent of dynamin-mediated endocytosis but regulated by the glypican members of heparan sulfate proteoglycans. Cell 119, 231–244 - PubMed
-
- Billuart P., Winter C. G., Maresh A., Zhao X., Luo L. (2001). Regulating axon branch stability: the role of p190 RhoGAP in repressing a retraction signaling pathway. Cell 107, 195–207 - PubMed
-
- Bloor J. W., Kiehart D. P. (2002). Drosophila RhoA regulates the cytoskeleton and cell-cell adhesion in the developing epidermis. Development 129, 3173–3183 - PubMed
-
- Casanueva M. O., Ferguson E. L. (2004). Germline stem cell number in the Drosophila ovary is regulated by redundant mechanisms that control Dpp signaling. Development 131, 1881–1890 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

