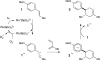Radical cation Diels-Alder cycloadditions by visible light photocatalysis
- PMID: 22032252
- PMCID: PMC3227774
- DOI: 10.1021/ja2093579
Radical cation Diels-Alder cycloadditions by visible light photocatalysis
Abstract
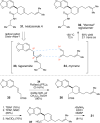
Ruthenium(II) polypyridyl complexes promote the efficient radical cation Diels-Alder cycloaddition of electron-rich dienophiles upon irradiation with visible light. These reactions enable facile [4 + 2] cycloadditions that would be electronically mismatched under thermal conditions. Key to the success of this methodology is the availability of ligand-modified ruthenium complexes that enable rational tuning of the electrochemical properties of the catalyst without significantly perturbing the overall photophysical properties of the system.
© 2011 American Chemical Society
Figures
References
-
- Bear BR, Sparks SM, Shea KJ. Angew. Chem. Int. Ed. 2001;40:820–849. - PubMed
- Nicolaou KC, Snyder SA, Montagnon T, Vassilikogiannakis G. Angew. Chem. Int. Ed. 2002;41:1668–1698. - PubMed
- Stocking EM, Williams RM. Angew. Chem. Int. Ed. 2003;42:3078–3115. - PubMed
- Takao K-I, Munakata R, Tadano K-I. Chem. Rev. 2005;105:4779–4807. - PubMed
-
- Bauld NL, Bellville DJ, Pabon R, Chelsky R, Green G. J. Am. Chem. Soc. 1983;105:2378–2382.
- Bellville DJ, Bauld NL, Pabon R, Gardner SA. J. Am. Chem. Soc. 1983;105:3584–3588.
- Pabon RA, Bauld NL. J. Am. Chem. Soc. 1984;106:1145–1146.
- Wiest O, Steckhan E, Grein F. J. Org. Chem. 1992;57:4034–4037.
- Bauld NL, Yang J. Org. Lett. 1999;1:773–774. - PubMed
- Valley NA, Wiest O. J. Org. Chem. 2007;72:559–566. - PubMed
- Sevov CS, Wiest O. J. Org. Chem. 2008;73:7909–7915. - PubMed
-
- Bellville DJ, Wirth DW, Bauld NL. J. Am. Chem. Soc. 1981;103:718–720.
- Yueh W, Bauld NL. J. Phys. Org. Chem. 1996;9:529–538.
-
-
For seminal reports of organic PET sensitization of radical cation Diels–Alder, see: Maroulis AJ, Arnold DR. J. Chem. Soc., Chem. Commun. 1979:351–352.; Jones CR, Allman BJ, Mooring A, Spahic B. J. Am. Chem. Soc. 1983;105:652–654.; Pabon RA, Bellville DJ, Bauld NL. J. Am. Chem. Soc. 1983;105:5158–5159.; Calhoun GC, Schuster GB. J. Am. Chem. Soc. 1984;106:6870–6871.; Mlcoch J, Steckhan E. Angew. Chem. Int. Ed. Eng. 1985;24:412–414.; Gieseler A, Steckhan E, Wiest O. Synlett. 1990:275–277.; Gieseler A, Steckhan E, Wiest O, Knoch F. J. Org. Chem. 1991;56:1405–1411.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources