Phosphatidylglycerol suppresses influenza A virus infection
- PMID: 22052877
- PMCID: PMC3359948
- DOI: 10.1165/rcmb.2011-0194OC
Phosphatidylglycerol suppresses influenza A virus infection
Abstract
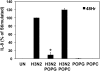
Influenza A virus (IAV) is a worldwide public health problem causing 500,000 deaths each year. Palmitoyl-oleoyl-phosphatidylglycerol (POPG) is a minor component of pulmonary surfactant, which has recently been reported to exert potent regulatory functions upon the innate immune system. In this article, we demonstrate that POPG acts as a strong antiviral agent against IAV. POPG markedly attenuated IL-8 production and cell death induced by IAV in cultured human bronchial epithelial cells. The lipid also suppressed viral attachment to the plasma membrane and subsequent replication in Madin-Darby canine kidney cells. Two virus strains, H1N1-PR8-IAV and H3N2-IAV, bind to POPG with high affinity, but exhibit only low-affinity interactions with the structurally related lipid, palmitoyl-oleoyl-phosphatidylcholine. Intranasal inoculation of H1N1-PR8-IAV in mice, in the presence of POPG, markedly suppressed the development of inflammatory cell infiltrates, the induction of IFN-γ recovered in bronchoalveolar lavage, and viral titers recovered from the lungs after 5 days of infection. These findings identify supplementary POPG as a potentially important new approach for treatment of IAV infections.
Figures






References
-
- Glezen WP. Clinical practice: prevention and treatment of seasonal influenza. N Engl J Med 2008;359:2579–2585 - PubMed
-
- Gooskens J, Jonges M, Claas E, Meijer A, van den Broek P, Kroes A. Morbidity and mortality associated with nosocomial transmission of oseltamivir-resistant influenza A (H1N1) virus. JAMA 2009;301:1066. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

