The mechanism of dynein motility: insight from crystal structures of the motor domain
- PMID: 22062687
- PMCID: PMC3249483
- DOI: 10.1016/j.bbamcr.2011.10.009
The mechanism of dynein motility: insight from crystal structures of the motor domain
Abstract
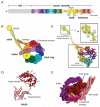
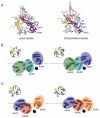
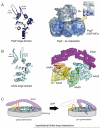
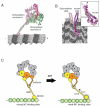
Dynein is a large cytoskeletal motor protein that belongs to the AAA+ (ATPases associated with diverse cellular activities) superfamily. While dynein has had a rich history of cellular research, its molecular mechanism of motility remains poorly understood. Here we describe recent X-ray crystallographic studies that reveal the architecture of dynein's catalytic ring, mechanical linker element, and microtubule binding domain. This structural information has given rise to new hypotheses on how the dynein motor domain might change its conformation in order to produce motility along microtubules.
Copyright © 2011 Elsevier B.V. All rights reserved.
Figures


References
-
- Gibbons IR, Rowe AJ. Dynein: A Protein with Adenosine Triphosphatase Activity from Cilia. Science. 1965;149:424–426. - PubMed
-
- Vallee RB, Williams JC, Varma D, Barnhart LE. Dynein: An ancient motor protein involved in multiple modes of transport. J Neurobiol. 2004;58:189–200. - PubMed
-
- Neuwald AF, Aravind L, Spouge JL, Koonin EV. AAA+: A class of chaperone-like ATPases associated with the assembly, operation, and disassembly of protein complexes. Genome Res. 1999;9:27–43. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

