Lower specific infectivity of protease-resistant prion protein generated in cell-free reactions
- PMID: 22065744
- PMCID: PMC3228482
- DOI: 10.1073/pnas.1111255108
Lower specific infectivity of protease-resistant prion protein generated in cell-free reactions
Abstract
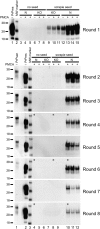
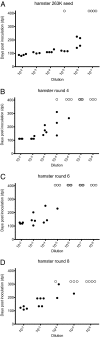
Prions are unconventional infectious agents that cause transmissible spongiform encephalopathy (TSE) diseases, or prion diseases. The biochemical nature of the prion infectious agent remains unclear. Previously, using a protein misfolding cyclic amplification (PMCA) reaction, infectivity and disease-associated protease-resistant prion protein (PrPres) were both generated under cell-free conditions, which supported a nonviral hypothesis for the agent. However, these studies lacked comparative quantitation of both infectivity titers and PrPres, which is important both for biological comparison with in vivo-derived infectivity and for excluding contamination to explain the results. Here during four to eight rounds of PMCA, end-point dilution titrations detected a >320-fold increase in infectivity versus that in controls. These results provide strong support for the hypothesis that the agent of prion infectivity is not a virus. PMCA-generated samples caused the same clinical disease and neuropathology with the same rapid incubation period as the input brain-derived scrapie samples, providing no evidence for generation of a new strain in PMCA. However, the ratio of the infectivity titer to the amount of PrPres (specific infectivity) was much lower in PMCA versus brain-derived samples, suggesting the possibility that a substantial portion of PrPres generated in PMCA might be noninfectious.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Comment in
-
Viruses do replicate in cell-free systems.Proc Natl Acad Sci U S A. 2012 Feb 21;109(8):E461; author reply E462. doi: 10.1073/pnas.1118908109. Epub 2012 Jan 30. Proc Natl Acad Sci U S A. 2012. PMID: 22308429 Free PMC article. No abstract available.
Similar articles
-
Generation of genuine prion infectivity by serial PMCA.Vet Microbiol. 2007 Aug 31;123(4):346-57. doi: 10.1016/j.vetmic.2007.04.004. Epub 2007 Apr 7. Vet Microbiol. 2007. PMID: 17493773
-
Ultra-efficient PrP(Sc) amplification highlights potentialities and pitfalls of PMCA technology.PLoS Pathog. 2011 Nov;7(11):e1002370. doi: 10.1371/journal.ppat.1002370. Epub 2011 Nov 17. PLoS Pathog. 2011. PMID: 22114554 Free PMC article.
-
In vitro generation of infectious scrapie prions.Cell. 2005 Apr 22;121(2):195-206. doi: 10.1016/j.cell.2005.02.011. Cell. 2005. PMID: 15851027
-
Prion protein conversion in vitro.J Mol Med (Berl). 2004 Jun;82(6):348-56. doi: 10.1007/s00109-004-0534-3. Epub 2004 Mar 10. J Mol Med (Berl). 2004. PMID: 15014886 Review.
-
Protein misfolding cyclic amplification (PMCA): Current status and future directions.Virus Res. 2015 Sep 2;207:47-61. doi: 10.1016/j.virusres.2014.11.007. Epub 2014 Nov 13. Virus Res. 2015. PMID: 25445341 Review.
Cited by
-
Role of the central lysine cluster and scrapie templating in the transmissibility of synthetic prion protein aggregates.PLoS Pathog. 2017 Sep 14;13(9):e1006623. doi: 10.1371/journal.ppat.1006623. eCollection 2017 Sep. PLoS Pathog. 2017. PMID: 28910420 Free PMC article.
-
Changes in prion replication environment cause prion strain mutation.FASEB J. 2013 Sep;27(9):3702-10. doi: 10.1096/fj.13-230466. Epub 2013 May 31. FASEB J. 2013. PMID: 23729586 Free PMC article.
-
Prions, proteinase K and infectivity.Prion. 2012 Nov-Dec;6(5):430-2. doi: 10.4161/pri.22309. Epub 2012 Oct 8. Prion. 2012. PMID: 23044510 Free PMC article.
-
Assessment of strain-specific PrP(Sc) elongation rates revealed a transformation of PrP(Sc) properties during protein misfolding cyclic amplification.PLoS One. 2012;7(7):e41210. doi: 10.1371/journal.pone.0041210. Epub 2012 Jul 17. PLoS One. 2012. PMID: 22815972 Free PMC article.
-
Rapid and Highly Sensitive Detection of Variant Creutzfeldt-Jakob Disease Abnormal Prion Protein on Steel Surfaces by Protein Misfolding Cyclic Amplification: Application to Prion Decontamination Studies.PLoS One. 2016 Jan 22;11(1):e0146833. doi: 10.1371/journal.pone.0146833. eCollection 2016. PLoS One. 2016. PMID: 26800081 Free PMC article.
References
-
- Chesebro B. Introduction to the transmissible spongiform encephalopathies or prion diseases. Br Med Bull. 2003;66:1–20. - PubMed
-
- Büeler H, et al. Mice devoid of PrP are resistant to scrapie. Cell. 1993;73:1339–1347. - PubMed
-
- Rohwer RG. The scrapie agent: “a virus by any other name”. In: Chesebro BW, editor. Transmissible Spongiform Encephalopathies, Scrapie, BSE and Related Disorders, Current Topics in Microbiology and Immunology. Vol. 172. New York: Springer; 1991. pp. 195–232. - PubMed
-
- Griffith JS. Self-replication and scrapie. Nature. 1967;215:1043–1044. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

