TGF-β mediates suppression of adipogenesis by estradiol through connective tissue growth factor induction
- PMID: 22067314
- PMCID: PMC3249674
- DOI: 10.1210/en.2011-1169
TGF-β mediates suppression of adipogenesis by estradiol through connective tissue growth factor induction
Abstract
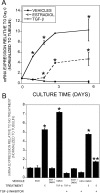
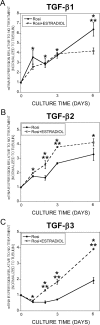
In the bone marrow cavity, adipocyte numbers increase, whereas osteoblast progenitor numbers decrease with aging. Because adipocytes and osteoblasts share a common progenitor, it is possible that this shift is due to an increase in adipocyte-lineage cells at the expense of osteoblast-lineage commitment. Estrogens inhibit adipocyte differentiation, and in both men and women, circulating estrogens correlate with bone loss with aging. In bone cells, estrogens stimulate expression of TGF-β and suppress mesenchymal cell adipogenesis. Using a tripotential mesenchymal cell line, we have examined whether estradiol suppression of adipocyte differentiation is due to stimulation of TGF-β and the mechanism by which TGF-β suppresses adipogenesis. We observed that estradiol-mediated suppression of adipogenic gene expression required at least 48 h treatment. TGF-β expression increased within 24 h of estradiol treatment, and TGF-β inhibition reversed estradiol influences on adipogenesis and adipocyte gene expression. Connective tissue growth factor (CTGF) mediates TGF-β suppression of adipogenesis in mouse 3T3-L1 cells. CTGF expression was induced within 24 h of TGF-β treatment, whereas estradiol-mediated induction required 48 h treatment. Moreover, estradiol-mediated induction of CTGF was abrogated by TGF-β inhibition. These data support that estradiol effects on adipogenesis involves TGF-β induction, which then induces CTGF to suppress adipogenesis.
Figures



Similar articles
-
Enamel matrix derivative inhibits adipocyte differentiation of 3T3-L1 cells via activation of TGF-βRI kinase activity.PLoS One. 2013 Aug 12;8(8):e71046. doi: 10.1371/journal.pone.0071046. eCollection 2013. PLoS One. 2013. PMID: 23951076 Free PMC article.
-
A new concept underlying stem cell lineage skewing that explains the detrimental effects of thiazolidinediones on bone.Stem Cells. 2010 May;28(5):916-27. doi: 10.1002/stem.405. Stem Cells. 2010. PMID: 20213769
-
TGF-β Signaling Plays an Essential Role in the Lineage Specification of Mesenchymal Stem/Progenitor Cells in Fetal Bone Marrow.Stem Cell Reports. 2019 Jul 9;13(1):48-60. doi: 10.1016/j.stemcr.2019.05.017. Epub 2019 Jun 13. Stem Cell Reports. 2019. PMID: 31204302 Free PMC article.
-
PPARγ and Wnt Signaling in Adipogenic and Osteogenic Differentiation of Mesenchymal Stem Cells.Curr Stem Cell Res Ther. 2016;11(3):216-25. doi: 10.2174/1574888x10666150519093429. Curr Stem Cell Res Ther. 2016. PMID: 25986621 Review.
-
The role of microRNAs in cell fate determination of mesenchymal stem cells: balancing adipogenesis and osteogenesis.BMB Rep. 2015 Jun;48(6):319-23. doi: 10.5483/bmbrep.2015.48.6.206. BMB Rep. 2015. PMID: 25341923 Free PMC article. Review.
Cited by
-
Transforming growth factor beta 1 induces CXCL16 and leukemia inhibitory factor expression in osteoclasts to modulate migration of osteoblast progenitors.Bone. 2013 Nov;57(1):68-75. doi: 10.1016/j.bone.2013.07.023. Epub 2013 Jul 25. Bone. 2013. PMID: 23891907 Free PMC article.
-
Androgens inhibit adipogenesis during human adipose stem cell commitment to preadipocyte formation.Steroids. 2013 Sep;78(9):920-6. doi: 10.1016/j.steroids.2013.05.001. Epub 2013 May 23. Steroids. 2013. PMID: 23707571 Free PMC article.
-
Association between Visceral and Bone Marrow Adipose Tissue and Bone Quality in Sedentary and Physically Active Ovariectomized Wistar Rats.Life (Basel). 2021 May 25;11(6):478. doi: 10.3390/life11060478. Life (Basel). 2021. PMID: 34070279 Free PMC article.
-
Function and Regulation of Bone Marrow Adipose Tissue in Health and Disease: State of the Field and Clinical Considerations.Compr Physiol. 2024 Jun 27;14(3):5521-5579. doi: 10.1002/cphy.c230016. Compr Physiol. 2024. PMID: 39109972 Free PMC article. Review.
-
Effects of sex and site on amino acid metabolism enzyme gene expression and activity in rat white adipose tissue.PeerJ. 2015 Nov 10;3:e1399. doi: 10.7717/peerj.1399. eCollection 2015. PeerJ. 2015. PMID: 26587356 Free PMC article.
References
-
- Riggs BL, Khosla S, Melton LJ., 3rd 2002. Sex steroids and the construction and conservation of the adult skeleton. Endocr Rev 23:279–302 - PubMed
-
- Spelsberg TC, Subramaniam M, Riggs BL, Khosla S. 1999. The actions and interactions of sex steroids and growth factors/cytokines on the skeleton. Mol Endocrinol 13:819–828 - PubMed
-
- Meunier P, Aaron J, Edouard C, Vignon G. 1971. Osteoporosis and the replacement of cell populations of the marrow by adipose tissue. A quantitative study of 84 iliac bone biopsies. Clin Orthop Relat Res 80:147–154 - PubMed
-
- Beresford JN, Bennett JH, Devlin C, Leboy PS, Owen ME. 1992. Evidence for an inverse relationship between the differentiation of adipocytic and osteogenic cells in rat marrow stromal cell cultures. J Cell Sci 102(Pt 2):341–351 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

