The role of mammalian target of rapamycin (mTOR) in the regulation of pancreatic β-cell mass: implications in the development of type-2 diabetes
- PMID: 22068611
- PMCID: PMC11114779
- DOI: 10.1007/s00018-011-0874-4
The role of mammalian target of rapamycin (mTOR) in the regulation of pancreatic β-cell mass: implications in the development of type-2 diabetes
Abstract
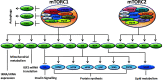
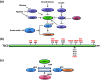
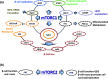
Type-2 diabetes mellitus (T2DM) is a disorder that is characterized by high blood glucose concentration in the context of insulin resistance and/or relative insulin deficiency. It causes metabolic changes that lead to the damage and functional impairment of organs and tissues resulting in increased morbidity and mortality. It is this form of diabetes whose prevalence is increasing at an alarming rate due to the 'obesity epidemic', as obesity is a key risk factor in the development of insulin resistance. However, the majority of individuals who have insulin resistance do not develop diabetes due to a compensatory increase in insulin secretion in response to an increase in insulin demand. This adaptive response is sustained by an increase in both β-cell function and mass. Importantly, there is increasing evidence that the Serine/Threonine kinase mammalian target of rapamycin (mTOR) plays a key role in the regulation of β-cell mass and therefore likely plays a critical role in β-cell adaptation. Therefore, the primary focus of this review is to summarize our current understanding of the role of mTOR in stimulating pancreatic β-cell mass and thus, in the prevention of type-2 diabetes.
Figures
References
-
- Ackermann AM, Gannon M. Molecular regulation of pancreatic beta-cell mass development, maintenance, and expansion. J Mol Endocrinol. 2007;38:193–206. - PubMed
-
- Alessi DR, James SR, Downes CP, Holmes AB, Gaffney PR, Reese CB, Cohen P. Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Balpha. Curr Biol. 1997;7:261–269. - PubMed
-
- Bai X, Ma D, Liu A, Shen X, Wang QJ, Liu Y, Jiang Y. Rheb activates mTOR by antagonizing its endogenous inhibitor, FKBP38. Science. 2007;318:977–980. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

