Hippocampal GluA1-containing AMPA receptors mediate context-dependent sensitization to morphine
- PMID: 22072679
- PMCID: PMC3235051
- DOI: 10.1523/JNEUROSCI.3835-11.2011
Hippocampal GluA1-containing AMPA receptors mediate context-dependent sensitization to morphine
Abstract
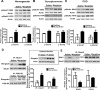
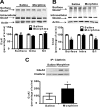
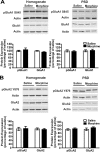
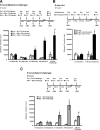
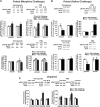
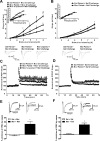
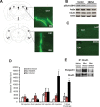
Glutamatergic systems, including AMPA receptors (AMPARs), are involved in opiate-induced neuronal and behavioral plasticity, although the mechanisms underlying these effects are not fully understood. In the present study, we investigated the effects of repeated morphine administration on AMPAR expression, synaptic plasticity, and context-dependent behavioral sensitization to morphine. We found that morphine treatment produced changes of synaptic AMPAR expression in the hippocampus, a brain area that is critically involved in learning and memory. These changes could be observed 1 week after the treatment, but only when mice developed context-dependent behavioral sensitization to morphine in which morphine treatment was associated with drug administration environment. Context-dependent behavioral sensitization to morphine was also associated with increased basal synaptic transmission and disrupted hippocampal long-term potentiation (LTP), whereas these effects were less robust when morphine administration was not paired with the drug administration environment. Interestingly, some effects may be related to the prior history of morphine exposure in the drug-associated environment, since alterations of AMPAR expression, basal synaptic transmission, and LTP were observed in mice that received a saline challenge 1 week after discontinuation of morphine treatment. Furthermore, we demonstrated that phosphorylation of GluA1 AMPAR subunit plays a critical role in the acquisition and expression of context-dependent behavioral sensitization, as this behavior is blocked by a viral vector that disrupts GluA1 phosphorylation. These data provide evidence that glutamatergic signaling in the hippocampus plays an important role in context-dependent sensitization to morphine and supports further investigation of glutamate-based strategies for treating opiate addiction.
Figures
References
-
- Billa SK, Liu J, Bjorklund NL, Sinha N, Fu Y, Shinnick-Gallagher P, Morón JA. Increased insertion of glutamate receptor 2-lacking alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) receptors at hippocampal synapses upon repeated morphine administration. Mol Pharmacol. 2010;77:874–883. - PMC - PubMed
-
- Borgkvist A, Valjent E, Santini E, Hervé D, Girault JA, Fisone G. Delayed, context- and dopamine D1 receptor-dependent activation of ERK in morphine-sensitized mice. Neuropharmacology. 2008;55:230–237. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
