Prominin-1 localizes to the open rims of outer segment lamellae in Xenopus laevis rod and cone photoreceptors
- PMID: 22076989
- PMCID: PMC3292372
- DOI: 10.1167/iovs.11-8635
Prominin-1 localizes to the open rims of outer segment lamellae in Xenopus laevis rod and cone photoreceptors
Abstract
Purpose: Prominin-1 expresses in rod and cone photoreceptors. Mutations in the prominin-1 gene cause retinal degeneration in humans. In this study, the authors investigated the expression and subcellular localization of xlProminin-1 protein, the Xenopus laevis ortholog of prominin-1, in rod and cone photoreceptors of this frog.
Methods: Antibodies specific for xlProminin-1 were generated. Immunoblotting was used to study the expression and posttranslational processing of xlProminin-1 protein. Immunocytochemical light and electron microscopy and transgenesis were used to study the subcellular distribution of xlProminin-1.
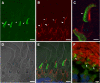
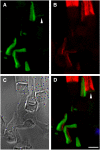
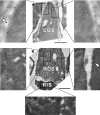
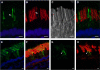
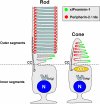
Results: xlProminin-1 is expressed and is subject to posttranslational proteolytic processing in the retina, brain, and kidney. xlProminin-1 is differently expressed and localized in outer segments of rod and cone photoreceptors of X. laevis. Antibodies specific for the N or C termini of xlProminin-1 labeled the open rims of lamellae of cone outer segments (COS) and the open lamellae at the base of rod outer segments (ROS). By contrast, anti-peripherin-2/rds antibody, Xper5A11, labeled the closed rims of cone lamellae adjacent to the ciliary axoneme and the rims of the closed ROS disks. The extent of labeling of the basal ROS by anti-xlProminin-1 antibodies varied with the light cycle in this frog. The entire ROS was also faintly labeled by both antibodies, a result that contrasts with the current notion that prominin-1 localizes only to the basal ROS.
Conclusions: These findings suggest that xlProminin-1 may serve as an anti-fusogenic factor in the regulation of disk morphogenesis and may help to maintain the open lamellar structure of basal ROS and COS disks in X. laevis photoreceptors.
Figures





References
-
- Young RW. Passage of newly formed protein through the connecting cilium of retina rods in the frog. J Ultrastruct Res. 1968;23:462–473 - PubMed
-
- Papermaster DS, Schneider BG, Besharse JC. Vesicular transport of newly synthesized opsin from the Golgi apparatus toward the rod outer segment: ultrastructural immunocytochemical and autoradiographic evidence in Xenopus retinas. Invest Ophthalmol Vis Sci. 1985;26:1386–1404 - PubMed
-
- Roepman R, Wolfrum U. Protein networks and complexes in photoreceptor cilia. Subcell Biochem. 2007;43:209–235 - PubMed
-
- Yau KW. Phototransduction mechanism in retinal rods and cones: the Friedenwald Lecture. Invest Ophthalmol Vis Sci. 1994;35:9–32 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

