Quantification of Thiopurine/UVA-Induced Singlet Oxygen Production
- PMID: 22081749
- PMCID: PMC3210580
- DOI: 10.1016/j.jphotochem.2011.09.001
Quantification of Thiopurine/UVA-Induced Singlet Oxygen Production
Abstract
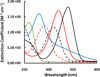
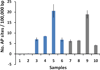
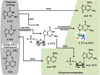
Thiopurines were examined for their ability to produce singlet oxygen ((1)O(2)) with UVA light. The target compounds were three thiopurine prodrugs, azathioprine (Aza), 6-mercaptopurine (6-MP) and 6-thioguanine (6-TG), and their S-methylated derivatives of 6-methylmercaptopurine (me6-MP) and 6-methylthioguanine (me6-TG). Our results showed that these thiopurines were efficient (1)O(2) sensitizers under UVA irradiation but rapidly lost their photoactivities for (1)O(2) production over time by a self-sensitized photooxidation of sulfur atoms in the presence of oxygen and UVA light. The initial quantum yields of (1)O(2) production were determined to be in the range of 0.30-0.6 in aqueous solutions. Substitution of a hydrogen atom with a nitroimidazole or methyl group at S decreased the efficacy of photosensitized (1)O(2) production as found for Aza, me6-MP and me6-TG. (1)O(2)-induced formation of 8-oxo-7,8-dihydro-2'-dexyguanosine (8-oxodGuo) was assessed by incubation of 6-methylthiopurine/UVA-treated calf thymus DNA with human repair enzyme 8-oxodGuo DNA glycosylase (hOGG1), followed by apurinic (AP) site determination. Because more 8-oxodGuo was formed in Tris D(2)O than in Tris H(2)O, (1)O(2) is implicated as a key species in the reaction. These findings provided quantitative information on the photosensitization efficacy of thiopurines and to some extent revealed the correlations between photoactivity and phototoxicity.
Figures


Similar articles
-
Combination of azathioprine and UVA irradiation is a major source of cellular 8-oxo-7,8-dihydro-2'-deoxyguanosine.DNA Repair (Amst). 2008 Dec 1;7(12):1982-9. doi: 10.1016/j.dnarep.2008.08.007. Epub 2008 Oct 1. DNA Repair (Amst). 2008. PMID: 18793759
-
Direct observation and quantitative characterization of singlet oxygen in aqueous solution upon UVA excitation of 6-thioguanines.J Phys Chem B. 2011 Mar 3;115(8):1889-94. doi: 10.1021/jp109590t. Epub 2011 Feb 4. J Phys Chem B. 2011. PMID: 21294562 Free PMC article.
-
Intramolecular Charge Transfer in the Azathioprine Prodrug Quenches Intersystem Crossing to the Reactive Triplet State in 6-Mercaptopurine.Photochem Photobiol. 2022 May;98(3):617-632. doi: 10.1111/php.13513. Epub 2021 Sep 28. Photochem Photobiol. 2022. PMID: 34480764
-
UVA photosensitization of thiopurines and skin cancer in organ transplant recipients.Photochem Photobiol Sci. 2012 Jan;11(1):62-8. doi: 10.1039/c1pp05194f. Epub 2011 Aug 23. Photochem Photobiol Sci. 2012. PMID: 21860872 Review.
-
Innovating Thiopurine Therapeutic Drug Monitoring: A Systematic Review and Meta-Analysis on DNA-Thioguanine Nucleotides (DNA-TG) as an Inclusive Biomarker in Thiopurine Therapy.Clin Pharmacokinet. 2024 Aug;63(8):1089-1109. doi: 10.1007/s40262-024-01393-0. Epub 2024 Jul 20. Clin Pharmacokinet. 2024. PMID: 39031224 Free PMC article.
Cited by
-
MUTYH mediates the toxicity of combined DNA 6-thioguanine and UVA radiation.Oncotarget. 2015 Apr 10;6(10):7481-92. doi: 10.18632/oncotarget.3037. Oncotarget. 2015. PMID: 25638157 Free PMC article.
-
Dual Functionality of 6-Methylthioguanine: Synergistic Effects Enhancing the Photolability of DNA Nucleobases.JACS Au. 2024 Jan 23;4(2):441-453. doi: 10.1021/jacsau.3c00575. eCollection 2024 Feb 26. JACS Au. 2024. PMID: 38425924 Free PMC article.
-
An Emerging Visible-Light Organic-Inorganic Hybrid Perovskite for Photocatalytic Applications.Nanomaterials (Basel). 2020 Jan 7;10(1):115. doi: 10.3390/nano10010115. Nanomaterials (Basel). 2020. PMID: 31936181 Free PMC article.
-
Excited-State Dynamics of the Thiopurine Prodrug 6-Thioguanine: Can N9-Glycosylation Affect Its Phototoxic Activity?Molecules. 2017 Feb 28;22(3):379. doi: 10.3390/molecules22030379. Molecules. 2017. PMID: 28264514 Free PMC article.
-
Mechanism of Resveratrol Dimers Isolated from Grape Inhibiting 1O2 Induced DNA Damage by UHPLC-QTOF-MS2 and UHPLC-QQQ-MS2 Analyses.Biomedicines. 2021 Mar 8;9(3):271. doi: 10.3390/biomedicines9030271. Biomedicines. 2021. PMID: 33800477 Free PMC article.
References
-
- Aarbakke J, Janka-Schaub G, Elion GB. Thiopurine biology and pharmacology. Trends in Pharmacological Sciences. 1997;18:3–8. - PubMed
-
- Relling MV, Dervieux T. Pharmacogenetics and cancer therapy. Nature Reviews Cancer. 2001;1:99–108. - PubMed
-
- Karran P. Thiopurines, DNA damage, DNA repair and therapy-related cancer. Br. Med. Bull. 2006;79–80:153–170. - PubMed
-
- Penn I. Tumour incidence in human allograft recipients. Transpl. Proc. 1979;XI:1047–1051. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
