A meta-analysis of lamivudine for interruption of mother-to-child transmission of hepatitis B virus
- PMID: 22090789
- PMCID: PMC3214708
- DOI: 10.3748/wjg.v17.i38.4321
A meta-analysis of lamivudine for interruption of mother-to-child transmission of hepatitis B virus
Abstract
Aim: To determine the therapeutic effect of lamivudine in late pregnancy for the interruption of mother-to-child transmission (MTCT) of hepatitis B virus (HBV).
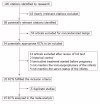
Methods: Studies were identified by searching available databases up to January 2011. Inclusive criteria were HBV-carrier mothers who had been involved in randomized controlled clinical trials (RCTs) with lamivudine treatment in late pregnancy, and newborns or infants whose serum hepatitis B surface antigen (HBsAg), hepatitis B e antigen (HBeAg) or HBV DNA had been documented. The relative risks (RRs) for interruption of MTCT as indicated by HBsAg, HBV DNA or HBeAg of newborns or infants were calculated with 95% confidence interval (CI) to estimate the efficacy of lamivudine treatment.
Results: Fifteen RCTs including 1693 HBV-carrier mothers were included in this meta-analysis. The overall RR was 0.43 (95% CI, 0.25-0.76; 8 RCTs; P(heterogeneity) = 0.04) and 0.33 (95% CI, 0.23-0.47; 6 RCTs; P(heterogeneity) = 0.93) indicated by newborn HBsAg or HBV DNA. The RR was 0.33 (95% CI, 0.21-0.50; 6 RCTs; P(heterogeneity) = 0.46) and 0.32 (95% CI, 0.20-0.50; 4 RCTs; P(heterogeneity) = 0.33) indicated by serum HBsAg or HBV DNA of infants 6-12 mo after birth. The RR (lamivudine vs hepatitis B immunoglobulin) was 0.27 (95% CI, 0.16-0.46; 5 RCTs; P(heterogeneity) = 0.94) and 0.24 (95% CI, 0.07-0.79; 3 RCTs; P(heterogeneity) = 0.60) indicated by newborn HBsAg or HBV DNA, respectively. In the mothers with viral load < 10⁶ copies/mL after lamivudine treatment, the efficacy (RR, 95% CI) was 0.33, 0.21-0.53 (5 RCTs; P(heterogeneity) = 0.82) for the interruption of MTCT, however, this value was not significant if maternal viral load was > 10⁶ copies/mL after lamivudine treatment (P = 0.45, 2 RCTs), as indicated by newborn serum HBsAg. The RR (lamivudine initiated from 28 wk of gestation vs control) was 0.34 (95% CI, 0.22-0.52; 7 RCTs; P(heterogeneity) = 0.92) and 0.33 (95% CI, 0.22-0.50; 5 RCTs; P(heterogeneity) = 0.86) indicated by newborn HBsAg or HBV DNA. The incidence of adverse effects of lamivudine was not higher in the mothers than in controls (P = 0.97). Only one study reported side effects of lamivudine in newborns.
Conclusion: Lamivudine treatment in HBV carrier-mothers from 28 wk of gestation may interrupt MTCT of HBV efficiently. Lamivudine is safe and more efficient than hepatitis B immunoglobulin in interrupting MTCT. HBV MTCT might be interrupted efficiently if maternal viral load is reduced to < 10⁶ copies/mL by lamivudine treatment.
Keywords: Efficacy; Hepatitis B virus; Lamivudine; Meta-analysis; Mother-to-child transmission.
Figures






References
-
- Lavanchy D. Worldwide epidemiology of HBV infection, disease burden, and vaccine prevention. J Clin Virol. 2005;34 Suppl 1:S1–S3. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

