Plasmodium falciparum parasites are killed by a transition state analogue of purine nucleoside phosphorylase in a primate animal model
- PMID: 22096507
- PMCID: PMC3214022
- DOI: 10.1371/journal.pone.0026916
Plasmodium falciparum parasites are killed by a transition state analogue of purine nucleoside phosphorylase in a primate animal model
Abstract
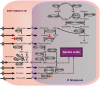
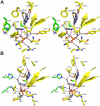
Plasmodium falciparum causes most of the one million annual deaths from malaria. Drug resistance is widespread and novel agents against new targets are needed to support combination-therapy approaches promoted by the World Health Organization. Plasmodium species are purine auxotrophs. Blocking purine nucleoside phosphorylase (PNP) kills cultured parasites by purine starvation. DADMe-Immucillin-G (BCX4945) is a transition state analogue of human and Plasmodium PNPs, binding with picomolar affinity. Here, we test BCX4945 in Aotus primates, an animal model for Plasmodium falciparum infections. Oral administration of BCX4945 for seven days results in parasite clearance and recrudescence in otherwise lethal infections of P. falciparum in Aotus monkeys. The molecular action of BCX4945 is demonstrated in crystal structures of human and P. falciparum PNPs. Metabolite analysis demonstrates that PNP blockade inhibits purine salvage and polyamine synthesis in the parasites. The efficacy, oral availability, chemical stability, unique mechanism of action and low toxicity of BCX4945 demonstrate potential for combination therapies with this novel antimalarial agent.
Conflict of interest statement
Figures

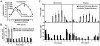
Similar articles
-
A resistant mutant of Plasmodium falciparum purine nucleoside phosphorylase uses wild-type neighbors to maintain parasite survival.J Biol Chem. 2021 Jan-Jun;296:100342. doi: 10.1016/j.jbc.2021.100342. Epub 2021 Jan 30. J Biol Chem. 2021. PMID: 33524395 Free PMC article.
-
Genetic resistance to purine nucleoside phosphorylase inhibition in Plasmodium falciparum.Proc Natl Acad Sci U S A. 2018 Feb 27;115(9):2114-2119. doi: 10.1073/pnas.1525670115. Epub 2018 Feb 12. Proc Natl Acad Sci U S A. 2018. PMID: 29440412 Free PMC article.
-
Purine-less death in Plasmodium falciparum induced by immucillin-H, a transition state analogue of purine nucleoside phosphorylase.J Biol Chem. 2002 Feb 1;277(5):3226-31. doi: 10.1074/jbc.M105906200. Epub 2001 Nov 12. J Biol Chem. 2002. PMID: 11706018
-
Targeting Plasmodium falciparum purine salvage enzymes: a look at structure-based drug development.Infect Disord Drug Targets. 2010 Jun;10(3):191-9. doi: 10.2174/187152610791163408. Infect Disord Drug Targets. 2010. PMID: 20480551 Review.
-
Immucillins in Infectious Diseases.ACS Infect Dis. 2018 Feb 9;4(2):107-117. doi: 10.1021/acsinfecdis.7b00172. Epub 2017 Dec 5. ACS Infect Dis. 2018. PMID: 29151351 Free PMC article. Review.
Cited by
-
Acyclic immucillin phosphonates: second-generation inhibitors of Plasmodium falciparum hypoxanthine-guanine-xanthine phosphoribosyltransferase.Chem Biol. 2012 Jun 22;19(6):721-30. doi: 10.1016/j.chembiol.2012.04.012. Chem Biol. 2012. PMID: 22726686 Free PMC article.
-
Inhibition and Mechanism of Plasmodium falciparum Hypoxanthine-Guanine-Xanthine Phosphoribosyltransferase.ACS Chem Biol. 2022 Dec 16;17(12):3407-3419. doi: 10.1021/acschembio.2c00546. Epub 2022 Nov 22. ACS Chem Biol. 2022. PMID: 36413975 Free PMC article.
-
Plasmodium falciparum finds the winning combination.J Biol Chem. 2021 Jan-Jun;296:100526. doi: 10.1016/j.jbc.2021.100526. Epub 2021 Apr 1. J Biol Chem. 2021. PMID: 34237898 Free PMC article.
-
Intracellular rebinding of transition-state analogues provides extended in vivo inhibition lifetimes on human purine nucleoside phosphorylase.J Biol Chem. 2017 Sep 22;292(38):15907-15915. doi: 10.1074/jbc.M117.801779. Epub 2017 Aug 9. J Biol Chem. 2017. PMID: 28794158 Free PMC article.
-
Alternatives to currently used antimalarial drugs: in search of a magic bullet.Infect Dis Poverty. 2016 Nov 4;5(1):103. doi: 10.1186/s40249-016-0196-8. Infect Dis Poverty. 2016. PMID: 27809883 Free PMC article. Review.
References
-
- Ting LM, Shi W, Lewandowicz A, Singh V, Mwakingwe A, et al. Targeting a novel Plasmodium falciparum purine recycling pathway with specific immucillins. J Biol Chem. 2005;280:9547–9554. - PubMed
-
- Downie MJ, Saliba KJ, Howitt SM, Broer S, Kirk K. Transport of nucleosides across the Plasmodium falciparum parasite plasma membrane has characteristics of PfENT1. Mol Microbiol. 2006;60:738–748. - PubMed
-
- Haider N, Eschbach ML, Dias Sde S, Gilberger TW, Walter RD, et al. The spermidine synthase of the malaria parasite Plasmodium falciparum: molecular and biochemical characterisation of the polyamine synthesis enzyme. Mol Biochem Parasitol. 2005;142:224–236. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

