The triterpenoid CDDO-Me promotes hematopoietic progenitor expansion and myelopoiesis in mice
- PMID: 22100978
- PMCID: PMC3502000
- DOI: 10.1016/j.bbmt.2011.11.013
The triterpenoid CDDO-Me promotes hematopoietic progenitor expansion and myelopoiesis in mice
Abstract
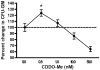
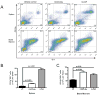
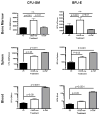
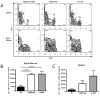
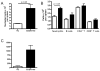
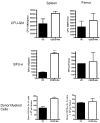
The synthetic triterpenoid CDDO-Me has been shown to directly inhibit the growth of myeloid leukemias and lends itself to a wide array of therapeutic indications, including inflammatory conditions, because of its inhibition of NF-κB. We have previously demonstrated protection from acute graft-versus-host disease after CDDO-Me administration in an allogeneic bone marrow transplantation model. In the current study, we observed that CDDO-Me promoted myelopoiesis in both naive and transplanted mice. This effect was dose dependent, as high doses of CDDO-Me inhibited myeloid growth in vitro. All lineages (granulocyte macrophage colony-forming unit, BFU-E) were promoted by CDDO-Me. We then compared the effects with granulocyte colony-stimulating factor, a known inducer of myeloid expansion and mobilization from the bone marrow. Whereas both drugs induced terminal myeloid expansion in the spleen, peripheral blood, and bone marrow, granulocyte colony-stimulating factor only induced granulocyte macrophage colony-forming unit precursors in the spleen, while CDDO-Me increased these precursors in the spleen and bone marrow. After sublethal total-body irradiation, mice pretreated with CDDO-Me further displayed an accelerated recovery of myeloid progenitors and total nucleated cells in the spleen. A similar expansion of myeloid and myeloid progenitors was noted with CDDO-Me treatment after syngeneic bone marrow transplantation. Combined, these data suggest that CDDO-Me may be of use posttransplantation to accelerate myeloid recovery in addition to the prevention of graft-versus-host disease.
Copyright © 2012 American Society for Blood and Marrow Transplantation. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
The remaining authors declare no competing financial interests.
Figures
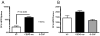
Similar articles
-
The triterpenoid CDDO-Me delays murine acute graft-versus-host disease with the preservation of graft-versus-tumor effects after allogeneic bone marrow transplantation.Biol Blood Marrow Transplant. 2010 Jun;16(6):739-50. doi: 10.1016/j.bbmt.2010.01.020. Epub 2010 Mar 23. Biol Blood Marrow Transplant. 2010. PMID: 20338256 Free PMC article.
-
Novel triterpenoid CDDO-Me is a potent inducer of apoptosis and differentiation in acute myelogenous leukemia.Blood. 2002 Jan 1;99(1):326-35. doi: 10.1182/blood.v99.1.326. Blood. 2002. PMID: 11756188
-
The synthetic triterpenoid, CDDO, suppresses alloreactive T cell responses and reduces murine early acute graft-versus-host disease mortality.Biol Blood Marrow Transplant. 2007 May;13(5):521-9. doi: 10.1016/j.bbmt.2006.12.453. Epub 2007 Feb 26. Biol Blood Marrow Transplant. 2007. PMID: 17448911 Free PMC article.
-
Roles of Hematopoietic Stem and Progenitor Cells in Ischemic Cardiovascular Disease.Curr Stem Cell Res Ther. 2021;16(5):589-598. doi: 10.2174/1574888X15666200130091858. Curr Stem Cell Res Ther. 2021. PMID: 32000654 Review.
-
The role of the microbiota in myelopoiesis during homeostasis and inflammation.Int Immunol. 2023 May 19;35(6):267-274. doi: 10.1093/intimm/dxad002. Int Immunol. 2023. PMID: 36694400 Free PMC article. Review.
Cited by
-
Bardoxolone methyl (CDDO-Me) as a therapeutic agent: an update on its pharmacokinetic and pharmacodynamic properties.Drug Des Devel Ther. 2014 Oct 23;8:2075-88. doi: 10.2147/DDDT.S68872. eCollection 2014. Drug Des Devel Ther. 2014. PMID: 25364233 Free PMC article. Review.
-
The triterpenoid RTA 408 is a robust mitigator of hematopoietic acute radiation syndrome in mice.Radiat Res. 2015 Mar;183(3):338-44. doi: 10.1667/RR13900.1. Epub 2015 Mar 4. Radiat Res. 2015. PMID: 25738896 Free PMC article.
-
Synthetic oleanane triterpenoids: multifunctional drugs with a broad range of applications for prevention and treatment of chronic disease.Pharmacol Rev. 2012 Oct;64(4):972-1003. doi: 10.1124/pr.111.004846. Epub 2012 Sep 10. Pharmacol Rev. 2012. PMID: 22966038 Free PMC article. Review.
-
Differential Sex Response to Aspirin in Decreasing Aneurysm Rupture in Humans and Mice.Hypertension. 2016 Aug;68(2):411-7. doi: 10.1161/HYPERTENSIONAHA.116.07515. Epub 2016 Jun 13. Hypertension. 2016. PMID: 27296993 Free PMC article.
References
-
- Einsele H, Bertz H, Beyer J, et al. Infectious complications after allogeneic stem cell transplantation: epidemiology and interventional therapy strategies--guidelines of the Infectious Diseases Working Party (AGIHO) of the German Society of Hematology and Oncology (DGHO) Annals of hematology. 2003;82(Suppl 2):S175–185. - PMC - PubMed
-
- Weaver CH, Schwartzberg LS, Hainsworth J, et al. Treatment-related mortality in 1000 consecutive patients receiving high-dose chemotherapy and peripheral blood progenitor cell transplantation in community cancer centers. Bone marrow transplantation. 1997;19:671–678. - PubMed
-
- Trivedi M, Martinez S, Corringham S, Medley K, Ball ED. Optimal use of G-CSF administration after hematopoietic SCT. Bone marrow transplantation. 2009;43:895–908. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

