Effect of a synbiotic yogurt on levels of fecal bifidobacteria, clostridia, and enterobacteria
- PMID: 22101054
- PMCID: PMC3272998
- DOI: 10.1128/AEM.05848-11
Effect of a synbiotic yogurt on levels of fecal bifidobacteria, clostridia, and enterobacteria
Abstract
While ingestion of synbiotic yogurts containing Bifidobacterium animalis subsp. lactis and inulin is increasing, their effect on certain microbial groups in the human intestine is unclear. To further investigate this, a large-scale, crossover-design, placebo-controlled study was utilized to evaluate the effect of a synbiotic yogurt containing B. animalis subsp. lactis Bb-12 and inulin on the human intestinal bifidobacteria, clostridia, and enterobacteria. Fecal samples were collected at 14 time points from 46 volunteers who completed the study, and changes in the intestinal bacterial levels were monitored using real-time PCR. Strain Bb-12 could not be detected in feces after 2 weeks of washout. A live/dead PCR procedure indicated that the Bb-12 strain detected in the fecal samples was alive. A significant increase (P < 0.001) in the total bifidobacterial numbers was seen in both groups of subjects during the final washout period compared to the prefeeding period. This increase in total bifidobacteria corresponded with a significant decrease (P < 0.05) in numbers of clostridia but not enterobacteria. No significant differences in numbers of bifidobacteria, clostridia, or enterobacteria were observed between the probiotic and placebo groups during any of the feeding periods. However, subgrouping subjects based on lower initial bifidobacterial numbers or higher initial clostridial numbers did show corresponding significant differences between the synbiotic yogurt and placebo groups. This was not observed for a subgroup with higher initial enterobacterial numbers. While this synbiotic yogurt can increase bifidobacterial numbers and decrease clostridial numbers (but not enterobacterial numbers) in some individuals, it cannot modulate these microbial groups in the majority of individuals.
Figures





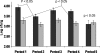


References
-
- Alander M, et al. 2001. Effect of galacto-oligosaccharide supplementation on human faecal microflora and on survival and persistence of Bifidobacterium lactis Bb-12 in the gastrointestinal tract. Int. Dairy J. 11:817–825
-
- Barnes EM, Ingram M. 1956. The effect of redox potential on the grown Clostridium welchii strain isolated from horse muscle. J. Appl. Bacteriol. 19:177–178
-
- Bartosch S, Woodmansey EJ, Paterson JCM, McMurdo MET, Macfarlane GT. 2005. Microbiological effects of consuming a synbiotic containing Bifidobacterium bifidum, Bifidobacterium lactis, and oligofructose in elderly persons, determined by real time polymerase chain reaction and counting of viable bacteria. Clin. Infect. Dis. 40:28–37 - PubMed
-
- Cai YM, Matsumoto M, Benno Y. 2000. Bifidobacterium lactis Meile et al. 1997 is a subjective synonym of Bifidobacterium animalis (Mitsuoka 1969) Scardovi and Trovatelli 1974. Microbiol. Immunol. 44:815–820 - PubMed
-
- Fallani M, et al. 2006. Clostridium difficile and Clostridium perfringens species detected in infant faecal microbiota using 16s rRNA targeted probes. J. Microbiol. Methods 67:150–161 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

