Differential gene expression in migratory streams of cortical interneurons
- PMID: 22103416
- PMCID: PMC3401901
- DOI: 10.1111/j.1460-9568.2011.07896.x
Differential gene expression in migratory streams of cortical interneurons
Abstract
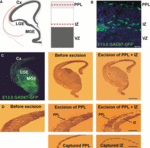
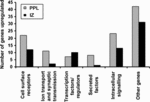
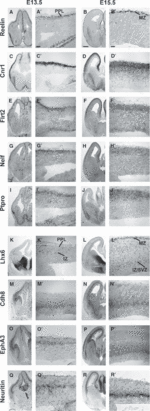
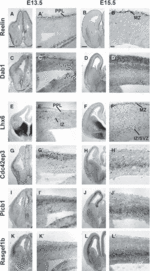
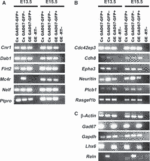
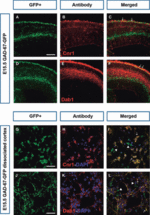
Cortical interneurons originate in the ganglionic eminences of the subpallium and migrate into the cortex in well-defined tangential streams. At the start of corticogenesis, two streams of migrating neurons are evident: a superficial one at the level of the preplate (PPL), and a deeper one at the level of the intermediate zone (IZ). Currently, little is known about the signalling mechanisms that regulate interneuron migration, and almost nothing is known about the molecules that may be involved in their choice of migratory stream. Here, we performed a microarray analysis, comparing the changes in gene expression between cells migrating in the PPL and those migrating in the IZ at embryonic day 13.5. This analysis identified genes, many of them novel, that were upregulated in one of the two streams. Moreover, polymerase chain reaction, in situ hybridization experiments and immunohistochemistry showed the expression of these genes in interneurons migrating within the PPL or IZ, suggesting that they play a role in their migration and choice of stream.
© 2011 The Authors. European Journal of Neuroscience © 2011 Federation of European Neuroscience Societies and Blackwell Publishing Ltd.
Figures
References
-
- Anderson SA, Marín O, Horn C, Jennings K, Rubenstein JL. Distinct cortical migrations from the medial and lateral ganglionic eminences. Development. 2001;128:353–363. - PubMed
-
- Andrews W, Liapi A, Plachez C, Camurri L, Zhang J, Mori S, Murakami F, Parnavelas JG, Sundaresan V, Richards LJ. Robo1 regulates the development of major axon tracts and interneuron migration in the forebrain. Development. 2006;133:2243–2252. - PubMed
-
- Andrews W, Barber M, Hernadez-Miranda LR, Xian J, Rakic S, Sundaresan V, Rabbitts TH, Pannell R, Rabbitts P, Thompson H, Erskine L, Murakami F, Parnavelas JG. The role of Slit–Robo signaling in the generation, migration and morphological differentiation of cortical interneurons. Dev. Biol. 2008;313:648–658. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

