Pregnancy-related effects on lamivudine pharmacokinetics in a population study with 228 women
- PMID: 22106227
- PMCID: PMC3264256
- DOI: 10.1128/AAC.00370-11
Pregnancy-related effects on lamivudine pharmacokinetics in a population study with 228 women
Abstract
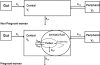
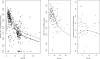
The aim of this study was to describe lamivudine (3TC) pharmacokinetics (PK) in HIV-infected nonpregnant and pregnant women and their fetuses. Samples were collected according to therapeutic drug monitoring from 228 women treated with lamivudine and retrospectively analyzed by a population approach. The samples were also collected from cord blood and amniotic fluid at birth. Lamivudine pharmacokinetics were ascribed to an open two-compartment model with linear absorption and elimination. Mean population parameter estimates (intersubject variability) for women were an absorption rate constant of 1.04 h(-1), an elimination clearance rate of 23.6 (0.266) liters · h(-1), a central volume of distribution of 109 (0.897) liters, an intercompartmental clearance rate of 6.7 liters/h, and a peripheral volume of distribution of 129 liters. A fetal compartment was linked to maternal circulation by mother-to-cord (or fetus) and cord-to-mother rate constants of 0.463 h(-1) and 0.538 h(-1), respectively. The amniotic fluid compartment was connected to the fetal compartment with an elimination rate constant of 0.163 h(-1) and a fixed-constant swallowing flow. The placental transfer expressed as fetal-to-maternal area under the concentration-time curve (AUC) ratio was 0.86, and the lamivudine amniotic fluid accumulation, expressed as the amniotic fluid-to-fetal AUC ratio, was 2.9. Pregnant women had a 22% higher apparent clearance than nonpregnant and parturient women; however, this increase did not lead to subexposure and should not require a dosage adjustment.
Figures


Similar articles
-
Pregnancy-related effects on tenofovir pharmacokinetics: a population study with 186 women.Antimicrob Agents Chemother. 2012 Feb;56(2):857-62. doi: 10.1128/AAC.05244-11. Epub 2011 Nov 28. Antimicrob Agents Chemother. 2012. PMID: 22123690 Free PMC article.
-
Maternal-fetal transfer and amniotic fluid accumulation of lamivudine in human immunodeficiency virus-infected pregnant women.Am J Obstet Gynecol. 2001 Jan;184(2):153-8. doi: 10.1067/mob.2001.108344. Am J Obstet Gynecol. 2001. PMID: 11174495
-
Population pharmacokinetics of emtricitabine in human immunodeficiency virus type 1-infected pregnant women and their neonates.Antimicrob Agents Chemother. 2009 Mar;53(3):1067-73. doi: 10.1128/AAC.00860-08. Epub 2008 Dec 22. Antimicrob Agents Chemother. 2009. PMID: 19104016 Free PMC article.
-
Pharmacokinetics of antiretroviral drugs in anatomical sanctuary sites: the fetal compartment (placenta and amniotic fluid).Antivir Ther. 2011;16(8):1139-47. doi: 10.3851/IMP1918. Antivir Ther. 2011. PMID: 22155898 Review.
-
Clinical pharmacokinetics of lamivudine.Clin Pharmacokinet. 1999 Jan;36(1):41-66. doi: 10.2165/00003088-199936010-00004. Clin Pharmacokinet. 1999. PMID: 9989342 Review.
Cited by
-
Obstetric Pharmacokinetic Dosing Studies are Urgently Needed.Front Pediatr. 2014 Feb 11;2:9. doi: 10.3389/fped.2014.00009. eCollection 2014. Front Pediatr. 2014. PMID: 24575394 Free PMC article. Review.
-
Role of Dolutegravir/Lamivudine in the Management of Pregnant People Living with HIV-1: A Narrative Review.Infect Dis Ther. 2025 Jan;14(1):59-80. doi: 10.1007/s40121-024-01085-z. Epub 2024 Dec 9. Infect Dis Ther. 2025. PMID: 39652285 Free PMC article. Review.
-
Prediction of Maternal and Fetal Acyclovir, Emtricitabine, Lamivudine, and Metformin Concentrations during Pregnancy Using a Physiologically Based Pharmacokinetic Modeling Approach.Clin Pharmacokinet. 2022 May;61(5):725-748. doi: 10.1007/s40262-021-01103-0. Epub 2022 Jan 24. Clin Pharmacokinet. 2022. PMID: 35067869
-
Physiologically-based pharmacokinetic modeling of renally excreted antiretroviral drugs in pregnant women.Br J Clin Pharmacol. 2015 Nov;80(5):1031-41. doi: 10.1111/bcp.12685. Epub 2015 Jul 22. Br J Clin Pharmacol. 2015. PMID: 26011128 Free PMC article.
-
Pregnancy-Associated Changes in Pharmacokinetics: A Systematic Review.PLoS Med. 2016 Nov 1;13(11):e1002160. doi: 10.1371/journal.pmed.1002160. eCollection 2016 Nov. PLoS Med. 2016. PMID: 27802281 Free PMC article.
References
-
- Althabe O, et al. 1976. Transference of para-amino-hippurate from the mother to the amniotic fluid. J. Perinat. Med. 4:227–233 - PubMed
-
- Anderson GD. 2005. Pregnancy-induced changes in pharmacokinetics: a mechanistic-based approach. Clin. Pharmacokinet. 44:989–1008 - PubMed
-
- Beal SL. 2001. Ways to fit a PK model with some data below the quantification limit. J. Pharmacokinet. Pharmacodyn. 28:481–504 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

