Differential regional expression patterns of α-synuclein, TNF-α, and IL-1β; and variable status of dopaminergic neurotoxicity in mouse brain after Paraquat treatment
- PMID: 22112368
- PMCID: PMC3247140
- DOI: 10.1186/1742-2094-8-163
Differential regional expression patterns of α-synuclein, TNF-α, and IL-1β; and variable status of dopaminergic neurotoxicity in mouse brain after Paraquat treatment
Abstract
Background: Paraquat (1, 1-dimethyl-4, 4-bipyridium dichloride; PQ) causes neurotoxicity, especially dopaminergic neurotoxicity, and is a supposed risk factor for Parkinson's disease (PD). However, the cellular and molecular mechanisms of PQ-induced neurodegeneration are far from clear. Previous studies have shown that PQ induces neuroinflammation and dopaminergic cell loss, but the prime cause of those events is still in debate.
Methods: We examined the neuropathological effects of PQ not only in substantia nigra (SN) but also in frontal cortex (FC) and hippocampus of the progressive mouse (adult Swiss albino) model of PD-like neurodegeneration, using immunohistochemistry, western blots, and histological and biochemical analyses.
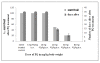
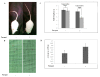
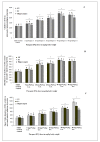
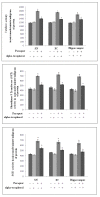
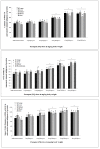
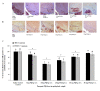
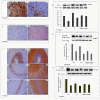
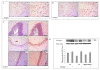
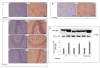
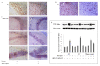
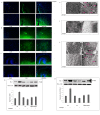
Results: PQ caused differential patterns of changes in cellular morphology and expression of proteins related to PD and neuroinflammation in the three regions examined (SN, FC and hippocampus). Coincident with behavioral impairment and brain-specific ROS generation, there was differential immunolocalization and decreased expression levels of tyrosine hydroxylase (TH) in the three regions, whereas α-synuclein immunopositivity increased in hippocampus, increased in FC and decreased in SN. PQ-induced neuroinflammation was characterized by area-specific changes in localization and appearances of microglial cells with or without activation and increment in expression patterns of tumor necrosis factor-α in the three regions of mouse brain. Expression of interleukin-1β was increased in FC and hippocampus but not significantly changed in SN.
Conclusion: The present study demonstrates that PQ induces ROS production and differential α-synuclein expression that promotes neuroinflammation in microglia-dependent or -independent manners, and produces different patterns of dopaminergic neurotoxicity in three different regions of mouse brain.
Figures


References
-
- Niso-Santano M, González-Polo RA, Bravo-San Pedro JM, Gómez-Sánchez R, Lastres-Becker I, Ortiz-Ortiz MA, Soler G, Morán JM, Cuadrado A, Fuentes JM. Activation of apoptosis signal-regulating kinase 1 is a key factor in paraquat-induced cell death: Modulation by the Nrf2/Trx axis. Free Rad Biol Med. 2010;48:1370–1381. doi: 10.1016/j.freeradbiomed.2010.02.024. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

