Allergenic lipid transfer proteins from plant-derived foods do not immunologically and clinically behave homogeneously: the kiwifruit LTP as a model
- PMID: 22114713
- PMCID: PMC3219694
- DOI: 10.1371/journal.pone.0027856
Allergenic lipid transfer proteins from plant-derived foods do not immunologically and clinically behave homogeneously: the kiwifruit LTP as a model
Abstract
Background: Food allergy is increasingly common worldwide. Tools for allergy diagnosis measuring IgE improved much since allergenic molecules and microarrays started to be used. IgE response toward allergens belonging to the same group of molecules has not been comprehensively explored using such approach yet.
Objective: Using the model of lipid transfer proteins (LTPs) from plants as allergens, including two new structures, we sought to define how heterogeneous is the behavior of homologous proteins.
Methods: Two new allergenic LTPs, Act d 10 and Act c 10, have been identified in green (Actinidia deliciosa) and gold (Actinidia chinensis) kiwifruit (KF), respectively, using clinically characterized allergic patients, and their biochemical features comparatively evaluated by means of amino acid sequence alignments. Along with other five LTPs from peach, mulberry, hazelnut, peanut, mugwort, KF LTPs, preliminary tested positive for IgE, have been immobilized on a microarray, used for IgE testing 1,003 allergic subjects. Comparative analysis has been carried out.
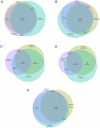
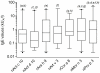
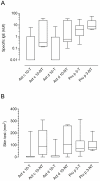
Results: Alignment of Act d 10 primary structure with the other allergenic LTPs shows amino acid identities to be in a narrow range between 40 and 55%, with a number of substitutions making the sequences quite different from each other. Although peach LTP dominates the IgE immune response in terms of prevalence, epitope recognition driven by sequence heterogeneity has been recorded to be distributed in a wide range of behaviors. KF LTPs IgE positive results were obtained in a patient subset IgE positive for the peach LTP. Anyhow, the negative results on homologous molecules allowed us to reintroduce KF in patients' diet.
Conclusion: The biochemical nature of allergenic molecule belonging to a group of homologous ones should not be taken as proof of immunological recognition as well. The availability of panels of homologous molecules to be tested using microarrays is valuable to address the therapeutic intervention.
Conflict of interest statement
Figures










References
-
- Prescott S, Allen KJ. Food allergy: Riding the second wave of the allergy epidemic. Pediatr Allergy Immunol. 2011;22:155–60. - PubMed
-
- Johansson SG, Bieber T, Dahl R, Friedmann PS, Lanier BQ, et al. Revised nomenclature for allergy for global use: Report of the Nomenclature Review Committee of the World Allergy Organization, October 2003. J Allergy Clin Immunol. 2004;113:832–6. - PubMed
-
- Mari A, Alessandri C, Bernardi ML, Ferrara R, Scala E, et al. Microarrayed Allergen Molecules for the Diagnosis of Allergic Diseases. Curr Allergy Asthma Rep. 2010;10:357–64. - PubMed
-
- Scala E, Alessandri C, Bernardi ML, Ferrara R, Palazzo P, et al. Cross-sectional survey on immunoglobulin E reactivity in 23 077 subjects using an allergenic molecule-based microarray detection system. Clin Exp Allergy. 2010;40:911–21. - PubMed
-
- Borges JP, Barre A, Culerrier R, Granier C, Didier A, et al. Lipid transfer proteins from Rosaceae fruits share consensus epitopes responsible for their IgE-binding cross-reactivity. Biochem Biophys Res Commun. 2008;365:685–90. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

