Changes in cotherapies after initiation of disease-modifying antirheumatic drug therapy in patients with rheumatoid arthritis
- PMID: 22121511
- PMCID: PMC3227007
- DOI: 10.1002/acr.20550
Changes in cotherapies after initiation of disease-modifying antirheumatic drug therapy in patients with rheumatoid arthritis
Abstract
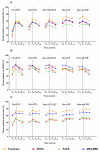
Objective. We hypothesized that initiation of a new disease-modifying antirheumatic drug (DMARD) for treatment of rheumatoid arthritis (RA) would decrease the use of corticosteroids, nonsteroidal antiinflammatory drugs (NSAIDs), and narcotics.Methods. Using administrative databases, we assembled 4 retrospective cohorts of RA patients (1998-2005) and identified 5 groups initiating DMARD regimens: methotrexate (MTX) with (new MTX) or without (first MTX) use of other nonbiologic DMARDs in the previous year; new hydroxychloroquine (HCQ) and/or sulfasalazine (SSZ; new HCQ/SSZ)and new leflunomide (new LEF), both with previous use of MTX; and new tumor necrosis factor α (TNFα) antagonists(new anti-TNF). We compared within-person differences in any use of cotherapies (≥ prescription) between the 6 months before and the 6-12 months after DMARD initiation.Results. Among 32,476 DMARD initiators, the prevalence of corticosteroid, NSAID, and narcotic use increased by 15%, 5%,and 6%, respectively, in the 6 months before initiation compared to the previous 6 months, suggesting worsening of the disease. In the 6-12 months after initiation for most initiator groups, more patients stopped using corticosteroids and NSAIDs than started, with overall decreases of 8.9% (95% confidence interval [95% CI] 8.4-9.4%) for corticosteroids and 12.9% (95%CI 12.3-13.4%) for NSAIDs. The proportion of narcotic users changed little (overall decrease of 2.5%; 95% CI 1.9-3.0%).Conclusion. Use of all 3 cotherapies increased in the 6 months before initiation of new DMARD regimens for RA. Use of corticosteroids and NSAIDs decreased modestly 6-12 months after initiation, but there was only a very small decrease in narcotic use. These differential changes require further study.
Figures

References
-
- Goldring SR. Pathogenesis of bone and cartilage destruction in rheumatoid arthritis. Rheumatology. 2003;42(suppl_2):ii11–ii16. - PubMed
-
- Guidelines for the management of rheumatoid arthritis: 2002 Update. Arthritis Rheum. 2002;46(2):328–346. - PubMed
-
- Quinn MA, Conaghan PG, Emery P. The therapeutic approach of early intervention for rheumatoid arthritis: what is the evidence? Rheumatology (Oxford) 2001;40(11):1211–1220. 5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

