Telmisartan ameliorates lipopolysaccharide-induced innate immune response through peroxisome proliferator-activated receptor-γ activation in human monocytes
- PMID: 22124178
- PMCID: PMC3237779
- DOI: 10.1097/HJH.0b013e32834dde5f
Telmisartan ameliorates lipopolysaccharide-induced innate immune response through peroxisome proliferator-activated receptor-γ activation in human monocytes
Abstract
Objective: Angiotensin II type 1 receptor (AT1) blockers (ARBs) reduce the bacterial endotoxin lipopolysaccharide (LPS)-induced innate immune response in human circulating monocytes expressing few AT1. To clarify the mechanisms of anti-inflammatory effects of ARBs with different peroxisome proliferator-activated receptor-γ (PPARγ)-activating potencies, we focused our study on telmisartan, an ARB with the highest PPARγ-stimulating activity.
Methods: Human circulating monocytes and monocytic THP-1 (human acute monocytic leukemia cell line) cells were exposed to 50 ng/ml LPS with or without pre-incubation with telmisartan. AT1 mRNA and protein expressions were determined by real-time PCR and membrane receptor binding assay, respectively. The expression of pro-inflammatory factors was determined by real-time PCR, western blot analysis and ELISA. PPARγ activation was measured by electrophoretic mobility shift assay and its role was determined by pharmacological inhibition and PPARγ gene silencing.
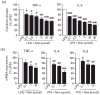
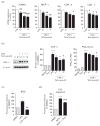
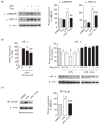
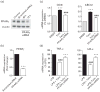
Results: In human monocytes, telmisartan significantly attenuated the LPS-induced expression of pro-inflammatory factors, the release of pro-inflammatory cytokines and prostaglandin E2, nuclear factor-κB activation and reactive oxygen species formation. In THP-1 cells, telmisartan significantly reduced LPS-induced tumor necrosis factor-α, inhibitor of κB-α, monocyte chemotactic protein-1 (MCP-1) and lectin-like oxidized low-density lipoprotein receptor-1 gene expression and MCP-1-directed migration. Telmisartan also stimulated the expression of the PPARγ target genes cluster of differentiation 36 and ATP-binding cassette subfamily G member 1 in monocytes. The anti-inflammatory effects of telmisartan were prevented by both PPARγ antagonism and PPARγ gene silencing. Anti-inflammatory effects of ARBs correlated with their PPARγ agonist potency.
Conclusion: Our observations demonstrate that in human monocytes, ARBs inhibit the LPS-induced pro-inflammatory response to a major extent through the PPARγ activation pathway and may be beneficial for the treatment of cardiovascular and metabolic disorders in which inflammation plays a major role.
Conflict of interest statement
There are no conflicts of interest.
Figures

References
-
- Timmermans PB, Wong PC, Chiu AT, Herblin WF, Benfield P, Carini DJ, et al. Angiotensin II receptors and angiotensin II receptor antagonists. Pharmacol Rev. 1993;45:205–251. - PubMed
-
- Chrysant SG, Chrysant GS, Chrysant C, Shiraz M. The treatment of cardiovascular disease continuum: focus on prevention and RAS blockade. Curr Clin Pharmacol. 2010;5:89–95. - PubMed
-
- Bakris G. Are there effects of renin-angiotensin system antagonists beyond blood pressure control? Am J Cardiol. 2010;105:21A–29A. - PubMed
-
- Konstam MA, Neaton JD, Dickstein K, Drexler H, Komajda M, Martinez FA, et al. Effects of high-dose versus low-dose losartan on clinical outcomes in patients with heart failure (HEAAL study): a randomised, double-blind trial. Lancet. 2009;374:1840–1848. - PubMed
-
- Neldam S. Choosing an angiotensin-receptor blocker: blood pressure lowering, cardiovascular protection or both? Future Cardiol. 2010;6:129–135. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

