Pigs, poultry, and pandemic influenza: how zoonotic pathogens threaten human health
- PMID: 22125035
- PMCID: PMC7123639
- DOI: 10.1007/978-1-4614-0204-6_6
Pigs, poultry, and pandemic influenza: how zoonotic pathogens threaten human health
Abstract
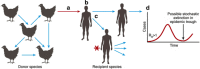
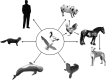
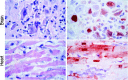
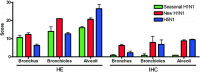
Emerging infections have an enormous impact on human health, food supply, economics, and the environment. Animals, and wild animals in particular, are considered to be the source of more than 70% of all emerging infections in humans. Two zoonotic influenza viruses that successfully spread from animals to humans are highly pathogenic avian influenza (HPAI) H5N1 virus, originating from poultry, and pandemic H1N1 influenza (pH1N1) virus, thought to originate from domestic swine. In this review, we discuss our research on the host species barrier for these viruses, concentrating on three questions: how does HPAI H5N1 virus transmit from birds to humans; what are the within-host dynamics of HPAI H5N1 virus and pH1N1 virus in humans and other mammals; and what determines transmission of influenza viruses among humans. To contain and eradicate zoonotic influenza viruses requires not only strategic virus surveillance of both animal and human populations, but also a better understanding of the hurdles that such a virus needs to jump over in order to cross the species barrier and cause a human pandemic. Advances in these two areas will allow us to better predict the risk of emergence of zoonotic influenza viruses in the human population.
Figures

References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

