Increased survival of glioblastoma patients who respond to antiangiogenic therapy with elevated blood perfusion
- PMID: 22127927
- PMCID: PMC3261301
- DOI: 10.1158/0008-5472.CAN-11-2464
Increased survival of glioblastoma patients who respond to antiangiogenic therapy with elevated blood perfusion
Erratum in
- Cancer Res. 2012 Jan 15;72(2):doi:10.1158/0008-5472.CAN-12-0113
- Cancer Res. 2012 Mar 1;72(5):1316
Abstract
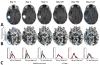
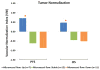
The abnormal vasculature of the tumor microenvironment supports progression and resistance to treatment. Judicious application of antiangiogenic therapy may normalize the structure and function of the tumor vasculature, promoting improved blood perfusion. However, direct clinical evidence is lacking for improvements in blood perfusion after antiangiogenic therapy. In this study, we used MRI to assess tumor blood perfusion in 30 recurrent glioblastoma patients who were undergoing treatment with cediranib, a pan-VEGF receptor tyrosine kinase inhibitor. Tumor blood perfusion increased durably for more than 1 month in 7 of 30 patients, in whom it was associated with longer survival. Together, our findings offer direct clinical evidence in support of the hypothesis that vascular normalization can increase tumor perfusion and help improve patient survival.
Trial registration: ClinicalTrials.gov NCT00035656.
Conflict of interest statement
Conflicts of interest
Figures


References
-
- Friedman HS, Prados MD, Wen PY, Mikkelsen T, Schiff D, Abrey LE, et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol. 2009;27:4733–40. - PubMed
-
- Jain RK. Normalizing tumor vasculature with anti-angiogenic therapy: a new paradigm for combination therapy. Nat Med. 2001;7:987–9. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- S10RR023043/RR/NCRR NIH HHS/United States
- S10 RR019254/RR/NCRR NIH HHS/United States
- P01CA80124/CA/NCI NIH HHS/United States
- P01 CA080124/CA/NCI NIH HHS/United States
- R01CA129371/CA/NCI NIH HHS/United States
- S10RR021110/RR/NCRR NIH HHS/United States
- S10RR019254/RR/NCRR NIH HHS/United States
- S10 RR023401/RR/NCRR NIH HHS/United States
- R01CA137254/CA/NCI NIH HHS/United States
- 5R01NS060918/NS/NINDS NIH HHS/United States
- R01 CA137254/CA/NCI NIH HHS/United States
- S10 RR021110/RR/NCRR NIH HHS/United States
- S10 RR019307/RR/NCRR NIH HHS/United States
- K24CA125440/CA/NCI NIH HHS/United States
- R21 CA117079/CA/NCI NIH HHS/United States
- R01 CA129371/CA/NCI NIH HHS/United States
- S10RR023401/RR/NCRR NIH HHS/United States
- K24 CA125440/CA/NCI NIH HHS/United States
- M01 RR001066/RR/NCRR NIH HHS/United States
- R21CA117079/CA/NCI NIH HHS/United States
- UL1 RR025758/RR/NCRR NIH HHS/United States
- S10RR019307/RR/NCRR NIH HHS/United States
- R01 NS060918/NS/NINDS NIH HHS/United States
- S10 RR023043/RR/NCRR NIH HHS/United States
- M01-RR-01066/RR/NCRR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

