Programmed cell death occurs asymmetrically during abscission in tomato
- PMID: 22128123
- PMCID: PMC3246325
- DOI: 10.1105/tpc.111.092494
Programmed cell death occurs asymmetrically during abscission in tomato
Abstract
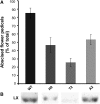
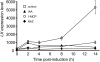
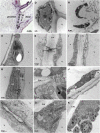
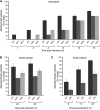
Abscission occurs specifically in the abscission zone (AZ) tissue as a natural stage of plant development. Previously, we observed delay of tomato (Solanum lycopersicum) leaf abscission when the LX ribonuclease (LX) was inhibited. The known association between LX expression and programmed cell death (PCD) suggested involvement of PCD in abscission. In this study, hallmarks of PCD were identified in the tomato leaf and flower AZs during the late stage of abscission. These included loss of cell viability, altered nuclear morphology, DNA fragmentation, elevated levels of reactive oxygen species and enzymatic activities, and expression of PCD-associated genes. Overexpression of antiapoptotic proteins resulted in retarded abscission, indicating PCD requirement. PCD, LX, and nuclease gene expression were visualized primarily in the AZ distal tissue, demonstrating an asymmetry between the two AZ sides. Asymmetric expression was observed for genes associated with cell wall hydrolysis, leading to AZ, or associated with ethylene biosynthesis, which induces abscission. These results suggest that different abscission-related processes occur asymmetrically between the AZ proximal and distal sides. Taken together, our findings identify PCD as a key mechanism that occurs asymmetrically during normal progression of abscission and suggest an important role for LX in this PCD process.
Figures





Similar articles
-
The pattern of 1-aminocyclopropane-1-carboxylate oxidase induction in the tomato leaf petiole abscission zone is independent of expression of the ribonuclease-LX-encoding LeLX gene.Plant Biol (Stuttg). 2018 Jul;20(4):722-728. doi: 10.1111/plb.12730. Epub 2018 Apr 26. Plant Biol (Stuttg). 2018. PMID: 29633546 Free PMC article.
-
Abscission of flowers and floral organs is closely associated with alkalization of the cytosol in abscission zone cells.J Exp Bot. 2015 Mar;66(5):1355-68. doi: 10.1093/jxb/eru483. Epub 2014 Dec 10. J Exp Bot. 2015. PMID: 25504336 Free PMC article.
-
Suppression of LX ribonuclease in tomato results in a delay of leaf senescence and abscission.Plant Physiol. 2006 Oct;142(2):710-21. doi: 10.1104/pp.106.080135. Epub 2006 Aug 18. Plant Physiol. 2006. PMID: 16920876 Free PMC article.
-
IDA: a peptide ligand regulating cell separation processes in Arabidopsis.J Exp Bot. 2013 Dec;64(17):5253-61. doi: 10.1093/jxb/ert338. Epub 2013 Oct 22. J Exp Bot. 2013. PMID: 24151306 Review.
-
Development and regulation of pedicel abscission in tomato.Front Plant Sci. 2015 Jun 11;6:442. doi: 10.3389/fpls.2015.00442. eCollection 2015. Front Plant Sci. 2015. PMID: 26124769 Free PMC article. Review.
Cited by
-
Burst of reactive oxygen species in pedicel-mediated fruit abscission after carbohydrate supply was cut off in longan (Dimocarpus longan).Front Plant Sci. 2015 May 26;6:360. doi: 10.3389/fpls.2015.00360. eCollection 2015. Front Plant Sci. 2015. PMID: 26074931 Free PMC article.
-
The pattern of 1-aminocyclopropane-1-carboxylate oxidase induction in the tomato leaf petiole abscission zone is independent of expression of the ribonuclease-LX-encoding LeLX gene.Plant Biol (Stuttg). 2018 Jul;20(4):722-728. doi: 10.1111/plb.12730. Epub 2018 Apr 26. Plant Biol (Stuttg). 2018. PMID: 29633546 Free PMC article.
-
Histological Staining and Hydrogen Peroxide Visualization of the Abscission Zone in Setaria viridis.Methods Mol Biol. 2025;2916:61-71. doi: 10.1007/978-1-0716-4470-6_6. Methods Mol Biol. 2025. PMID: 40366586
-
Phytohormonal dynamics in the abscission zone of Korla fragrant pear during calyx abscission: a visual study.Front Plant Sci. 2024 Oct 8;15:1452072. doi: 10.3389/fpls.2024.1452072. eCollection 2024. Front Plant Sci. 2024. PMID: 39439514 Free PMC article.
-
SlERF52 regulates SlTIP1;1 expression to accelerate tomato pedicel abscission.Plant Physiol. 2021 Apr 23;185(4):1829-1846. doi: 10.1093/plphys/kiab026. Plant Physiol. 2021. PMID: 33638643 Free PMC article.
References
-
- Aleksandrushkina N.I., Vanyushin B.F. (2009). Endonucleases and their involvement in plant apoptosis. Russ. J. Plant Physiol. 56: 291–305
-
- An Q., Ehlers K., Kogel K.H., van Bel A.J.E., Hückelhoven R. (2006). Multivesicular compartments proliferate in susceptible and resistant MLA12-barley leaves in response to infection by the biotrophic powdery mildew fungus. New Phytol. 172: 563–576 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources

