The role of β-arrestin2 in the mechanism of morphine tolerance in the mouse and guinea pig gastrointestinal tract
- PMID: 22129596
- PMCID: PMC3286318
- DOI: 10.1124/jpet.111.186320
The role of β-arrestin2 in the mechanism of morphine tolerance in the mouse and guinea pig gastrointestinal tract
Abstract
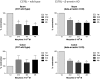
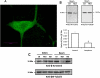
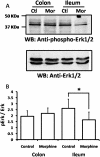
β-Arrestin2 has been reported to play an essential role in analgesic tolerance. Analgesic tolerance without concomitant tolerance to constipation is a limiting side effect of chronic morphine treatment. Because tolerance to morphine develops in the mouse ileum but not the colon, we therefore examined whether the role of β-arrestin2 in the mechanism of morphine tolerance differs in the ileum and colon. In both guinea pig and mouse, chronic in vitro exposure (2 h, 10 μM) to morphine resulted in tolerance development in the isolated ileum but not the colon. The IC(50) values for morphine-induced inhibition of electrical field stimulation contraction of guinea pig longitudinal muscle myenteric plexus shifted rightward in the ileum from 5.7 ± 0.08 (n = 9) to 5.45 ± 0.09 (n = 6) (p < 0.001) after morphine exposure. A significant shift was not observed in the colon. Similar differential tolerance was seen between the mouse ileum and the colon. However, tolerance developed in the colon from β-arrestin2 knockout mice. β-Arrestin2 and extracellular signal-regulated kinase 1/2 expression levels were determined further by Western blot analyses in guinea pig longitudinal muscle myenteric plexus. A time-dependent decrease in the expression of β-arrestin2 and extracellular signal-regulated kinase 1/2 occurred in the ileum but not the colon after 2 h of morphine (10 μM) exposure. Naloxone prevented the decrease in β-arrestin2. In the isolated ileum from guinea pigs chronically treated in vivo with morphine for 7 days, neither additional tolerance to in vitro exposure of morphine nor a decrease in β-arrestin2 occurred. We conclude that a decrease in β-arrestin2 is associated with tolerance development to morphine in the gastrointestinal tract.
Figures
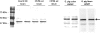




References
-
- Bailey CP, Smith FL, Kelly E, Dewey WL, Henderson G. (2006) How important is protein kinase C in mu-opioid receptor desensitization and morphine tolerance? Trends Pharmacol Sci 27:558–565 - PubMed
-
- Bohn LM, Gainetdinov RR, Lin FT, Lefkowitz RJ, Caron MG. (2000) Mu-opioid receptor desensitization by beta-arrestin-2 determines morphine tolerance but not dependence. Nature 408:720–723 - PubMed
-
- Bohn LM, Lefkowitz RJ, Gainetdinov RR, Peppel K, Caron MG, Lin FT. (1999) Enhanced morphine analgesia in mice lacking beta-arrestin 2. Science 286:2495–2498 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

