Mice with asthma are more resistant to influenza virus infection and NK cells activated by the induction of asthma have potentially protective effects
- PMID: 22134539
- PMCID: PMC3305878
- DOI: 10.1007/s10875-011-9619-2
Mice with asthma are more resistant to influenza virus infection and NK cells activated by the induction of asthma have potentially protective effects
Abstract
Purpose: This study was conducted in order to investigate whether the virulence of the influenza virus infection is affected by asthma in mice.
Methods: Mice with asthma or control mice were infected with influenza virus. The survival rate, body weight, virus titer, cytokine profile, and cell infiltration in bronchoalveolar lavage fluid (BALF) were measured. The NK cell cytotoxicity was determined by a co-culture system with YAC-1 cells, and the effects of NK cells were observed by depletion of NK cells using anti-asialoGM1 serum. The virus-specific CD8(+) T cell killing assay was also performed.
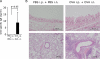
Results: When asthmatic or control mice were infected with non- and sub-lethal doses of influenza virus, the asthmatic mice were more resistant to the virus than control mice with regard to the survival rate, the remission of body weight loss, and the virus burden. Anti-viral cytokines and the NK cell number were increased in the BALF of asthmatic mice before the infection. The NK cell cytotoxicity in the asthmatic mice was significantly enhanced compared to that in control mice, and the depletion of NK cells in asthmatic mice was abrogated both the improved survival rate and the recovery of the body weight loss. The antigen-specific CD8(+) T cell killing activity in asthmatic mice was also significantly increased following the infection compared to that in control mice.
Conclusion: NK cell activated by the induction of asthma and the subsequently activated antigen-specific CD8(+) T cells could promptly eliminate the viral-infected cells, thus leading to improvements in the morbidity and mortality of influenza virus infection.
Figures




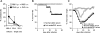

Similar articles
-
Asthma increases susceptibility to heterologous but not homologous secondary influenza.J Virol. 2014 Aug;88(16):9166-81. doi: 10.1128/JVI.00265-14. Epub 2014 Jun 4. J Virol. 2014. PMID: 24899197 Free PMC article.
-
IFN-γ production downstream of NKT cell activation in mice infected with influenza virus enhances the cytolytic activities of both NK cells and viral antigen-specific CD8+ T cells.Virology. 2010 Nov 25;407(2):325-32. doi: 10.1016/j.virol.2010.08.030. Epub 2010 Sep 19. Virology. 2010. PMID: 20855097
-
Type I interferon plays opposing roles in cytotoxicity and interferon-γ production by natural killer and CD8 T cells after influenza A virus infection in mice.J Innate Immun. 2014;6(4):456-66. doi: 10.1159/000356824. Epub 2014 Jan 10. J Innate Immun. 2014. PMID: 24435166 Free PMC article.
-
NK cells regulate CD8+ T cell priming and dendritic cell migration during influenza A infection by IFN-γ and perforin-dependent mechanisms.J Immunol. 2012 Sep 1;189(5):2099-109. doi: 10.4049/jimmunol.1103474. Epub 2012 Aug 6. J Immunol. 2012. PMID: 22869906
-
Phenotypic and Functional Characteristics of a Novel Influenza Virus Hemagglutinin-Specific Memory NK Cell.J Virol. 2021 May 24;95(12):e00165-21. doi: 10.1128/JVI.00165-21. Print 2021 May 24. J Virol. 2021. PMID: 33827945 Free PMC article.
Cited by
-
Impact of Therapeutics on Unified Immunity During Allergic Asthma and Respiratory Infections.Front Allergy. 2022 Mar 25;3:852067. doi: 10.3389/falgy.2022.852067. eCollection 2022. Front Allergy. 2022. PMID: 35386652 Free PMC article. Review.
-
The immune profile associated with acute allergic asthma accelerates clearance of influenza virus.Immunol Cell Biol. 2014 May-Jun;92(5):449-59. doi: 10.1038/icb.2013.113. Epub 2014 Jan 28. Immunol Cell Biol. 2014. PMID: 24469764 Free PMC article.
-
Prevention of Influenza Virus-Induced Immunopathology by TGF-β Produced during Allergic Asthma.PLoS Pathog. 2015 Sep 25;11(9):e1005180. doi: 10.1371/journal.ppat.1005180. eCollection 2015 Sep. PLoS Pathog. 2015. PMID: 26407325 Free PMC article.
-
A paucigranulocytic asthma host environment promotes the emergence of virulent influenza viral variants.Elife. 2021 Feb 16;10:e61803. doi: 10.7554/eLife.61803. Elife. 2021. PMID: 33588989 Free PMC article.
-
Asthma increases susceptibility to heterologous but not homologous secondary influenza.J Virol. 2014 Aug;88(16):9166-81. doi: 10.1128/JVI.00265-14. Epub 2014 Jun 4. J Virol. 2014. PMID: 24899197 Free PMC article.
References
-
- Gilca R, De Serres G, Boulianne N, Ouhoummane N, Papenburg J, Douville-Fradet M, Fortin E, Dionne M, Boivin G, Skowronski DM. Risk factors for hospitalization and severe outcomes of 2009 pandemic H1N1 influenza in Quebec, Canada. Influenza Other Respi Viruses. 2011;5:247–255. doi: 10.1111/j.1750-2659.2011.00204.x. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

