Deafferentation-induced redistribution of MMP-2, but not of MMP-9, depends on the emergence of GAP-43 positive axons in the adult rat cochlear nucleus
- PMID: 22135757
- PMCID: PMC3202138
- DOI: 10.1155/2011/859359
Deafferentation-induced redistribution of MMP-2, but not of MMP-9, depends on the emergence of GAP-43 positive axons in the adult rat cochlear nucleus
Abstract
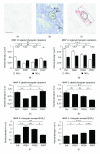
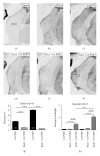
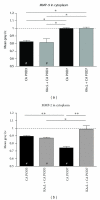
The matrix metalloproteinases MMP-9 and MMP-2, major modulators of the extracellular matrix (ECM), were changed in amount and distribution in the rat anteroventral cochlear nucleus (AVCN) following its sensory deafferentation by cochlear ablation. To determine what causal relationships exist between the redistribution of MMP-9 and MMP-2 and deafferentation-induced reinnervation, kainic acid was stereotaxically injected into the ventral nucleus of the trapezoid body (VNTB) prior to cochlear ablation, killing cells that deliver the growth associated protein 43 (GAP-43) into AVCN. Deafferentation-induced changes in the pattern of MMP-9 staining remained unaffected by VNTB lesions. By contrast, changes in the distribution of MMP-2 normally evoked by sensory deafferentation were reversed if GAP-43 positive axons were prevented to grow in AVCN. In conclusion, GAP-43-containing axons emerging in AVCN after cochlear ablation seem to be causal for the maintenance of MMP-2-mediated ECM remodeling.
Figures




Similar articles
-
Differential molecular profiles of astrocytes in degeneration and re-innervation after sensory deafferentation of the adult rat cochlear nucleus.Eur J Neurosci. 2013 Jul;38(1):2041-56. doi: 10.1111/ejn.12200. Epub 2013 Apr 15. Eur J Neurosci. 2013. PMID: 23581580
-
MMP-2 is involved in synaptic remodeling after cochlear lesion.Neuroreport. 2010 Mar 31;21(5):324-7. doi: 10.1097/WNR.0b013e328335b518. Neuroreport. 2010. PMID: 20173666
-
Synaptic reorganization in the adult rat's ventral cochlear nucleus following its total sensory deafferentation.PLoS One. 2011;6(8):e23686. doi: 10.1371/journal.pone.0023686. Epub 2011 Aug 22. PLoS One. 2011. PMID: 21887295 Free PMC article.
-
Superior olivary contributions to auditory system plasticity: medial but not lateral olivocochlear neurons are the source of cochleotomy-induced GAP-43 expression in the ventral cochlear nucleus.J Comp Neurol. 2004 Jul 26;475(3):374-90. doi: 10.1002/cne.20180. J Comp Neurol. 2004. PMID: 15221952
-
Matrix metalloproteinase-2 (MMP-2) and MMP-9 in pulmonary pathology.Exp Lung Res. 2005 Jul-Aug;31(6):599-621. doi: 10.1080/019021490944232. Exp Lung Res. 2005. PMID: 16019990 Review.
Cited by
-
Matrix Metalloproteinases During Axonal Regeneration, a Multifactorial Role from Start to Finish.Mol Neurobiol. 2017 Apr;54(3):2114-2125. doi: 10.1007/s12035-016-9801-x. Epub 2016 Feb 29. Mol Neurobiol. 2017. PMID: 26924318 Review.
-
Inhibition of MMP-2 but not MMP-9 influences inner ear spiral ganglion neurons in vitro.Cell Mol Neurobiol. 2014 Oct;34(7):1011-21. doi: 10.1007/s10571-014-0077-2. Epub 2014 Jun 17. Cell Mol Neurobiol. 2014. PMID: 24935409 Free PMC article.
-
Sensory deafferentation modulates and redistributes neurocan in the rat auditory brainstem.Brain Behav. 2019 Aug;9(8):e01353. doi: 10.1002/brb3.1353. Epub 2019 Jul 4. Brain Behav. 2019. PMID: 31271523 Free PMC article.
References
-
- Kane EC. Patterns of degeneration in the caudal cochlear nucleus of the cat after cochlear ablation. Anatomical Record. 1974;179(1):67–92. - PubMed
-
- Altschuler RA, Sheridan CE, Horn JW, Wenthold RJ. Immunocytochemical localization of glutamate immunoreactivity in the guinea pig cochlea. Hearing Research. 1989;42(2-3):167–174. - PubMed
-
- Illing R-B, Horváth M, Laszig R. Plasticity of the auditory brainstem: effects of cochlear ablation on GAP-43 immunoreactivity in the rat. Journal of Comparative Neurology. 1997;382(1):116–138. - PubMed
-
- Illing R-B, Kraus KS, Meidinger MA. Reconnecting neuronal networks in the auditory brainstem following unilateral deafening. Hearing Research. 2005;206(1-2):185–199. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

