Optical reversal of halothane-induced immobility in C. elegans
- PMID: 22137475
- PMCID: PMC3245814
- DOI: 10.1016/j.cub.2011.10.042
Optical reversal of halothane-induced immobility in C. elegans
Abstract
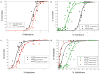
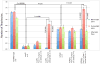
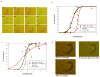
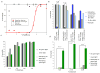
Volatile anesthetics (VAs) cause profound neurological effects, including reversible loss of consciousness and immobility. Despite their widespread use, the mechanism of action of VAs remains one of the unsolved puzzles of neuroscience [1, 2]. Genetic studies in Caenorhabditis elegans [3, 4], Drosophila [3, 5], and mice [6-9] indicate that ion channels controlling the neuronal resting membrane potential (RMP) also control anesthetic sensitivity. Leak channels selective for K(+) [10-13] or permeable to Na(+) [14] are critical for establishing RMP. We hypothesized that halothane, a VA, caused immobility by altering the neuronal RMP. In C. elegans, halothane-induced immobility is acutely and completely reversed by channelrhodopsin-2 based depolarization of the RMP when expressed specifically in cholinergic neurons. Furthermore, hyperpolarizing cholinergic neurons via halorhodopsin activation increases sensitivity to halothane. The sensitivity of C. elegans to halothane can be altered by 25-fold by either manipulation of membrane conductance with optogenetic methods or generation of mutations in leak channels that set the RMP. Immobility induced by another VA, isoflurane, is not affected by these treatments, thereby excluding the possibility of nonspecific hyperactivity. The sum of our data indicates that leak channels and the RMP are important determinants of halothane-induced general anesthesia.
Copyright © 2011 Elsevier Ltd. All rights reserved.
Figures
Comment in
-
Anesthetic mechanisms: worms light the way.Curr Biol. 2011 Dec 20;21(24):R985-6. doi: 10.1016/j.cub.2011.11.012. Curr Biol. 2011. PMID: 22192828
Similar articles
-
Anesthetic mechanisms: worms light the way.Curr Biol. 2011 Dec 20;21(24):R985-6. doi: 10.1016/j.cub.2011.11.012. Curr Biol. 2011. PMID: 22192828
-
Common genetic determinants of halothane and isoflurane potencies in Caenorhabditis elegans.Anesthesiology. 1998 Dec;89(6):1509-17. doi: 10.1097/00000542-199812000-00030. Anesthesiology. 1998. PMID: 9856727
-
Motoneuronal TASK channels contribute to immobilizing effects of inhalational general anesthetics.J Neurosci. 2010 Jun 2;30(22):7691-704. doi: 10.1523/JNEUROSCI.1655-10.2010. J Neurosci. 2010. PMID: 20519544 Free PMC article.
-
Tandem pore domain K channels: an important site of volatile anesthetic action?Curr Drug Targets. 2000 Sep;1(2):207-17. doi: 10.2174/1389450003349335. Curr Drug Targets. 2000. PMID: 11465071 Review.
-
A quantitative genetic approach towards volatile anesthetic mechanisms in C. elegans.Toxicol Lett. 1998 Nov 23;100-101:309-17. doi: 10.1016/s0378-4274(98)00200-8. Toxicol Lett. 1998. PMID: 10049158 Review.
Cited by
-
NALCN ion channels have alternative selectivity filters resembling calcium channels or sodium channels.PLoS One. 2013;8(1):e55088. doi: 10.1371/journal.pone.0055088. Epub 2013 Jan 28. PLoS One. 2013. PMID: 23383067 Free PMC article.
-
Demonstrating Connections Between Neuron Signaling and Behavior using C. elegans Learning Assays and Optogenetics in a Laboratory Class.J Undergrad Neurosci Educ. 2018 Sep 15;16(3):A223-A231. eCollection 2018 Summer. J Undergrad Neurosci Educ. 2018. PMID: 30254536 Free PMC article.
-
Drosophila ryanodine receptors mediate general anesthesia by halothane.Anesthesiology. 2013 Mar;118(3):587-601. doi: 10.1097/ALN.0b013e31827e52c6. Anesthesiology. 2013. PMID: 23254148 Free PMC article.
-
Sodium Leak Channel in Glutamatergic Neurons of the Lateral Parabrachial Nucleus Helps to Maintain Respiratory Frequency Under Sevoflurane Anesthesia.Neurosci Bull. 2024 Aug;40(8):1127-1140. doi: 10.1007/s12264-024-01223-0. Epub 2024 May 20. Neurosci Bull. 2024. PMID: 38767833 Free PMC article.
-
Novel activation of voltage-gated K(+) channels by sevoflurane.J Biol Chem. 2012 Nov 23;287(48):40425-32. doi: 10.1074/jbc.M112.405787. Epub 2012 Oct 4. J Biol Chem. 2012. PMID: 23038249 Free PMC article.
References
-
- Campagna JA, Miller KW, Forman SA. Mechanisms of actions of inhaled anesthetics. N Engl J Med. 2003;348:2110–2124. - PubMed
-
- Humphrey JA, Hamming KS, Thacker CM, Scott RL, Sedensky MM, Snutch TP, Morgan PG, Nash HA. A putative cation channel and its novel regulator: cross-species conservation of effects on general anesthesia. Curr Biol. 2007;17:624–629. - PubMed
-
- Morgan PG, Sedensky MM, Meneely PM, Cascorbi HF. The effect of two genes on anesthetic response in the nematode Caenorhabditis elegans. Anesth. 1988;69:246–251. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

