Activation of calpains mediates early lung neutrophilic inflammation in ventilator-induced lung injury
- PMID: 22140070
- PMCID: PMC3289265
- DOI: 10.1152/ajplung.00349.2011
Activation of calpains mediates early lung neutrophilic inflammation in ventilator-induced lung injury
Abstract
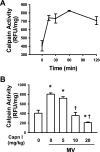
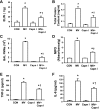
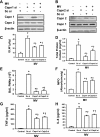
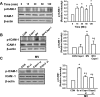
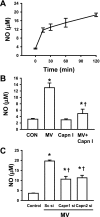
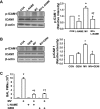
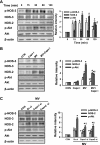
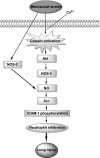
Lung inflammatory responses in the absence of infection are considered to be one of primary mechanisms of ventilator-induced lung injury. Here, we determined the role of calpain in the pathogenesis of lung inflammation attributable to mechanical ventilation. Male C57BL/6J mice were subjected to high (28 ml/kg) tidal volume ventilation for 2 h in the absence and presence of calpain inhibitor I (10 mg/kg). To address the isoform-specific functions of calpain 1 and calpain 2 during mechanical ventilation, we utilized a liposome-based delivery system to introduce small interfering RNAs targeting each isoform in pulmonary vasculature in vivo. Mechanical ventilation with high tidal volume induced rapid (within minutes) and persistent calpain activation and lung inflammation as evidenced by neutrophil recruitment, production of TNF-α and IL-6, pulmonary vascular hyperpermeability, and lung edema formation. Pharmaceutical calpain inhibition significantly attenuated these inflammatory responses caused by lung hyperinflation. Depletion of calpain 1 or calpain 2 had a protective effect against ventilator-induced lung inflammatory responses. Inhibition of calpain activity by means of siRNA silencing or pharmacological inhibition also reduced endothelial nitric oxide (NO) synthase (NOS-3)-mediated NO production and subsequent ICAM-1 phosphorylation following high tidal volume ventilation. These results suggest that calpain activation mediates early lung inflammation during ventilator-induced lung injury via NOS-3/NO-dependent ICAM-1 phosphorylation and neutrophil recruitment. Inhibition of calpain activation may therefore provide a novel and promising strategy for the prevention and treatment of ventilator-induced lung injury.
Figures
Similar articles
-
Ropivacaine attenuates endotoxin plus hyperinflation-mediated acute lung injury via inhibition of early-onset Src-dependent signaling.BMC Anesthesiol. 2014 Jul 19;14:57. doi: 10.1186/1471-2253-14-57. eCollection 2014. BMC Anesthesiol. 2014. PMID: 25097454 Free PMC article.
-
Activation of Src-dependent Smad3 signaling mediates the neutrophilic inflammation and oxidative stress in hyperoxia-augmented ventilator-induced lung injury.Respir Res. 2015 Sep 16;16(1):112. doi: 10.1186/s12931-015-0275-6. Respir Res. 2015. PMID: 26377087 Free PMC article.
-
Mechanical induction of group V phospholipase A(2) causes lung inflammation and acute lung injury.Am J Physiol Lung Cell Mol Physiol. 2013 May 15;304(10):L689-700. doi: 10.1152/ajplung.00047.2013. Epub 2013 Mar 22. Am J Physiol Lung Cell Mol Physiol. 2013. PMID: 23525785 Free PMC article.
-
Inhibition of HMGCoA reductase by simvastatin protects mice from injurious mechanical ventilation.Respir Res. 2015 Feb 14;16(1):24. doi: 10.1186/s12931-015-0173-y. Respir Res. 2015. PMID: 25848815 Free PMC article.
-
Heme oxygenase-1 system, inflammation and ventilator-induced lung injury.Eur J Pharmacol. 2012 Feb 29;677(1-3):1-4. doi: 10.1016/j.ejphar.2011.12.010. Epub 2011 Dec 16. Eur J Pharmacol. 2012. PMID: 22200625 Review.
Cited by
-
Enhancing Extracellular Adenosine Levels Restores Barrier Function in Acute Lung Injury Through Expression of Focal Adhesion Proteins.Front Mol Biosci. 2021 Mar 10;8:636678. doi: 10.3389/fmolb.2021.636678. eCollection 2021. Front Mol Biosci. 2021. PMID: 33778007 Free PMC article.
-
Ropivacaine attenuates endotoxin plus hyperinflation-mediated acute lung injury via inhibition of early-onset Src-dependent signaling.BMC Anesthesiol. 2014 Jul 19;14:57. doi: 10.1186/1471-2253-14-57. eCollection 2014. BMC Anesthesiol. 2014. PMID: 25097454 Free PMC article.
-
Critical role of calpain in inflammation.Biomed Rep. 2016 Dec;5(6):647-652. doi: 10.3892/br.2016.785. Epub 2016 Oct 19. Biomed Rep. 2016. PMID: 28101338 Free PMC article.
-
Activation of NLRP3 inflammasome in alveolar macrophages contributes to mechanical stretch-induced lung inflammation and injury.J Immunol. 2013 Apr 1;190(7):3590-9. doi: 10.4049/jimmunol.1200860. Epub 2013 Feb 22. J Immunol. 2013. PMID: 23436933 Free PMC article.
-
Differential role for p120-catenin in regulation of TLR4 signaling in macrophages.J Immunol. 2014 Aug 15;193(4):1931-41. doi: 10.4049/jimmunol.1302863. Epub 2014 Jul 11. J Immunol. 2014. PMID: 25015829 Free PMC article.
References
-
- Abraham E, Carmody A, Shenkar R, Arcaroli J. Neutrophils as early immunologic effectors in hemorrhage- or endotoxemia-induced acute lung injury. Am J Physiol Lung Cell Mol Physiol 279: L1137–L1145, 2000 - PubMed
-
- Acute Respiratory Distress Syndrome Network Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 342: 1301–1308, 2000 - PubMed
-
- Averna M, Stifanese R, De Tullio R, Salamino F, Bertuccio M, Pontremoli S, Melloni E. Proteolytic degradation of nitric oxide synthase isoforms by calpain is modulated by the expression levels of HSP90. FEBS J 274: 6116–6127, 2007 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

