Emerging technologies for making glycan-defined glycoproteins
- PMID: 22141574
- PMCID: PMC3262938
- DOI: 10.1021/cb200429n
Emerging technologies for making glycan-defined glycoproteins
Abstract
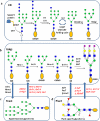
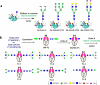
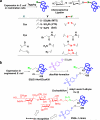
Protein glycosylation is a common and complex posttranslational modification of proteins, which expands functional diversity while boosting structural heterogeneity. Glycoproteins, the end products of such a modification, are typically produced as mixtures of glycoforms possessing the same polypeptide backbone but differing in the site of glycosylation and/or in the structures of pendant glycans, from which single glycoforms are difficult to isolate. The urgent need for glycan-defined glycoproteins in both detailed structure-function relationship studies and therapeutic applications has stimulated an extensive interest in developing various methods for manipulating protein glycosylation. This review highlights emerging technologies that hold great promise in making a variety of glycan-defined glycoproteins, with a particular emphasis in the following three areas: specific glycoengineering of host biosynthetic pathways, in vitro chemoenzymatic glycosylation remodeling, and chemoselective and site-specific glycosylation of proteins.
Figures


Similar articles
-
N-glycosylation engineering of biopharmaceutical expression systems.Curr Mol Med. 2009 Sep;9(7):774-800. doi: 10.2174/156652409789105552. Curr Mol Med. 2009. PMID: 19860659 Review.
-
Carbohydrate synthesis and biosynthesis technologies for cracking of the glycan code: recent advances.Biotechnol Adv. 2013 Jan-Feb;31(1):17-37. doi: 10.1016/j.biotechadv.2012.03.008. Epub 2012 Mar 28. Biotechnol Adv. 2013. PMID: 22484115 Review.
-
Glycoengineering Chinese hamster ovary cells: a short history.Biochem Soc Trans. 2021 Apr 30;49(2):915-931. doi: 10.1042/BST20200840. Biochem Soc Trans. 2021. PMID: 33704400 Free PMC article. Review.
-
Glycan Remodeling of Human Erythropoietin (EPO) Through Combined Mammalian Cell Engineering and Chemoenzymatic Transglycosylation.ACS Chem Biol. 2017 Jun 16;12(6):1665-1673. doi: 10.1021/acschembio.7b00282. Epub 2017 May 5. ACS Chem Biol. 2017. PMID: 28452462 Free PMC article.
-
Glycoengineering of Antibodies for Modulating Functions.Annu Rev Biochem. 2019 Jun 20;88:433-459. doi: 10.1146/annurev-biochem-062917-012911. Epub 2019 Mar 27. Annu Rev Biochem. 2019. PMID: 30917003 Free PMC article. Review.
Cited by
-
Chemical and chemoenzymatic synthesis of glycoproteins for deciphering functions.Chem Biol. 2014 Jan 16;21(1):51-66. doi: 10.1016/j.chembiol.2014.01.001. Chem Biol. 2014. PMID: 24439206 Free PMC article. Review.
-
Synthetic Glycobiology: Parts, Systems, and Applications.ACS Synth Biol. 2020 Jul 17;9(7):1534-1562. doi: 10.1021/acssynbio.0c00210. Epub 2020 Jun 30. ACS Synth Biol. 2020. PMID: 32526139 Free PMC article. Review.
-
Glycoconjugates: Synthesis, Functional Studies, and Therapeutic Developments.Chem Rev. 2022 Oct 26;122(20):15603-15671. doi: 10.1021/acs.chemrev.1c01032. Epub 2022 Sep 29. Chem Rev. 2022. PMID: 36174107 Free PMC article. Review.
-
Immune recruitment or suppression by glycan engineering of endogenous and therapeutic antibodies.Biochim Biophys Acta. 2016 Aug;1860(8):1655-68. doi: 10.1016/j.bbagen.2016.04.016. Epub 2016 Apr 20. Biochim Biophys Acta. 2016. PMID: 27105835 Free PMC article. Review.
-
Enterococcus faecalis α1-2-mannosidase (EfMan-I): an efficient catalyst for glycoprotein N-glycan modification.FEBS Lett. 2020 Feb;594(3):439-451. doi: 10.1002/1873-3468.13618. Epub 2019 Oct 8. FEBS Lett. 2020. PMID: 31552675 Free PMC article.
References
-
- Dwek RA. Glycobiology: Toward understanding the function of sugars. Chem. Rev. 1996;96:683–720. - PubMed
-
- Haltiwanger RS, Lowe JB. Role of glycosylation in development. Annu. Rev. Biochem. 2004;73:491–537. - PubMed
-
- Dube DH, Bertozzi CR. Glycans in cancer and inflammation--potential for therapeutics and diagnostics. Nat. Rev. Drug Discov. 2005;4:477–488. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

