Juvenile hormone action through a defined enhancer motif to modulate ecdysteroid-activation of natural core promoters
- PMID: 22142799
- PMCID: PMC3277835
- DOI: 10.1016/j.cbpb.2011.11.008
Juvenile hormone action through a defined enhancer motif to modulate ecdysteroid-activation of natural core promoters
Abstract
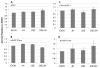
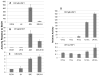
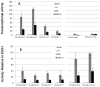
We have established a model system of hormone action, in an Sf9 cell transfection system, using defined enhancer motifs and natural core promoters of metamorphosis-associated genes. The DR1 enhancer, that is an established DNA binding site for the ecdysone receptor/ultraspiracle heterodimer, was necessary for transcriptional activation by 20-OH ecdysone. For this activated transcription, a natural sequence closely 5' to the TATA box is necessary. Cotreatment with juvenile hormone III strongly suppressed the steroid activation of transcription. However, in the absence of the sequence located closely 5' to the TATA box, cotreatment with juvenile hormone instead increased transcription over that occurring due to 20-hydroxy-ecdysone alone. This sensitivity to activation by cotreatment with juvenile hormone could be transferred to a related, but otherwise unresponsive, hexamerin core promoter simply by transferring to the unresponsive promoter the five base transcription start site (ACAGT) from the responsive hexamerin gene. These are the first reports that the direction of JH action on 20-OH ecdysone-activated transcription can be reversed by removal of a sequence at the core promoter, and that modulatory action of juvenile hormone can be transferred to a different gene by transferring the transcription start site motif.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures
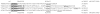
Similar articles
-
Interactions of ultraspiracle with ecdysone receptor in the transduction of ecdysone- and juvenile hormone-signaling.FEBS J. 2005 Apr;272(7):1577-89. doi: 10.1111/j.1742-4658.2005.04578.x. FEBS J. 2005. PMID: 15794746
-
Identification of regulatory sequences of juvenile hormone-sensitive and -insensitive serum protein-encoding genes.Gene. 1996 Sep 16;173(2):209-14. doi: 10.1016/0378-1119(96)00100-x. Gene. 1996. PMID: 8964501
-
Regulation of the juvenile hormone esterase gene by a composite core promoter.Biochem J. 2000 Feb 15;346 Pt 1(Pt 1):233-40. Biochem J. 2000. PMID: 10657262 Free PMC article.
-
The DPE, a core promoter element for transcription by RNA polymerase II.Exp Mol Med. 2002 Sep 30;34(4):259-64. doi: 10.1038/emm.2002.36. Exp Mol Med. 2002. PMID: 12515390 Review.
-
Hormone receptors and the regulation of insect metamorphosis.Receptor. 1993 Fall;3(3):203-9. Receptor. 1993. PMID: 8167571 Review.
Cited by
-
Large-scale analysis of Drosophila core promoter function using synthetic promoters.Mol Syst Biol. 2022 Feb;18(2):e9816. doi: 10.15252/msb.20209816. Mol Syst Biol. 2022. PMID: 35156763 Free PMC article.
-
Molecular Mechanisms of Transcription Activation by Juvenile Hormone: A Critical Role for bHLH-PAS and Nuclear Receptor Proteins.Insects. 2012 Mar 22;3(1):324-38. doi: 10.3390/insects3010324. Insects. 2012. PMID: 26467963 Free PMC article. Review.
-
Ecdysone signaling underlies the pea aphid transgenerational wing polyphenism.Proc Natl Acad Sci U S A. 2017 Feb 7;114(6):1419-1423. doi: 10.1073/pnas.1617640114. Epub 2017 Jan 23. Proc Natl Acad Sci U S A. 2017. PMID: 28115695 Free PMC article.
References
-
- Abdou MA, He Q, Wen D, Zyaan O, Wang J, Xu J, Baumann AA, Joseph J, Wilson TG, Li S, Wang J. Drosophila Met and Gce are partially redundant in transducing juvenile hormone action. Insect Biochem Mol Biol. 2011 Sep 29; [Epub ahead of print] - PubMed
-
- Berger EM, Goudie K, Klieger L, Berger M, DeCato R. The juvenile hormone analogue, methoprene, inhibits ecdysterone induction of small heat shock protein gene expression. Dev Biol. 1992;151:410–8. - PubMed
-
- Berger EM, Dubrovsky EB. Juvenile hormone molecular actions and interactions during development of Drosophila melanogaster. Vitam Horm. 2005;73:175–215. - PubMed
-
- Bitra K, Palli SR. Interaction of proteins involved in ecdysone and juvenile hormone signal transduction. Arch Insect Biochem Physiol. 2009;70:90–105. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

